
LNP Hand Mixing: Why LNP mixing methods matter, and Comparison with Microfluidic Mixing
Abstract
LNP hand mixing is widely used for RNA-LNP formulation due to its simplicity and minimal equipment requirements. However, the method introduces poorly controlled mixing kinetics, leading to intrinsic variability in nanoparticle size, limited reproducibility, and poor scalability, which may compromise translational validity. In this study, we directly compare hand mixing with microfluidic mixing using a clinically relevant ALC-0315 LNP formulation. Microfluidic preparation generated smaller and more homogeneous nanoparticles and resulted in substantially higher expression both in vitro and in vivo, despite comparable RNA encapsulation efficiency. These findings demonstrate that the mixing strategy alone can significantly alter RNA delivery outcomes and should therefore be considered a critical experimental parameter in RNA-LNP research and development.
Introduction on LNP formulation mixing strategies
Choosing an appropriate mixing strategy for lipid nanoparticle (LNP) formulation is far more than a technical detail. The mixing step directly governs key physicochemical characteristics—such as nanoparticle size, size distribution, and encapsulation efficiency, which in turn strongly influence the biological performance of RNA-LNPs both in vitro and in vivo.
Hand mixing is widely used in early-stage research due to its simplicity and accessibility, but it suffers from intrinsic limitations in batch-to-batch reproducibility, operator dependency, and scalability. These constraints become increasingly critical when moving from early stage screening studies to in vitro & in-vivo evaluation and translational development. In contrast, microfluidic mixing provides a higher level of control over nanoparticle formation by enabling rapid and well-defined mixing, leading to improved repeatability, more consistent LNP assembly, and enhanced biological performance.
In this short review, we present a data-driven comparison of hand mixing and microfluidic mixing using an ALC-0315 (Pfizer/BioNTech) lipid formulation with yeast RNA based on a study carried out with the team of Kurt Ristoph at Purdue University1 in partnership with Inside Therapeutics. Our results demonstrate how faster and more controlled microfluidic mixing leads to smaller nanoparticles, improved repeatability, and superior in vitro and in vivo performance, while remaining highly flexible across research and development scales.
LNP Mixing Methods: Control vs Variability
All current RNA-LNP synthesis processes rely on bottom-up assembly mechanisms, where an ethanolic lipid phase is mixed with an aqueous phase containing RNA in excess. This rapid solvent exchange triggers LNP formation through nanoprecipitation and self-assembly.
During this process, the final LNP characteristics are highly sensitive to the mixing conditions, as these directly govern the kinetics of nucleation and particle growth. In general, faster and more homogeneous mixing favors the formation of smaller nanoparticles, while reproducible mixing conditions are essential to ensure consistency across batches.
Several mixing strategies can be used to formulate LNPs. Here, we focus on two of the most commonly employed approaches: hand mixing and microfluidic mixing.
What is LNP hand mixing?
LNP hand mixing is a manual formulation method in which an ethanolic lipid solution is added to an aqueous RNA solution using a pipette, often combined with aspiration–dispense cycles or vortexing to for RNA-LNP
| Advantages of LNP hand mixing | Limitations of LNP hand mixing |
|---|---|
| Very accessible, no specialized equipment | Slow mixing times leading to larger particle sizes |
| Compatible with very low formulation volumes | Poorly defined due to local concentration gradients |
| Strong operator dependency | |
| Extremely limited scalability for in vitro and in vivo studies |
What is Microfluidics LNP formulation?
Microfluidic mixing operates under laminar flow conditions, where lipid and aqueous phases are combined within ms under continuous and well-defined hydrodynamic conditions. Key formulation parameters— including flow rate ratio (FRR) and total flow rate (TFR)—are precisely controlled, enabling tight regulation of the LNP formation process.
| Advantages of microfluidics for LNP formulation | Limitations of microfluidics |
|---|---|
| Much faster mixing kinetics, enabling smaller LNP formation | Requires dedicated microfluidic equipment |
| Excellent control and repeatability of mixing conditions | |
| Minimal operator dependency | |
| High flexibility from very low volumes to in vivo-relevant quantities |
As a result, microfluidic mixing consistently produces smaller and more uniform LNPs compared to bulk hand-mixing methods, while offering a scalable and translatable formulation strategy.
Practical comparison of hand-mixing and microfluidics for LNP formulation
To assess the practical differences between the two approaches, the same standard LNP formulation—based on the Pfizer/BioNTech ALC-0315 lipid composition—was prepared using both hand mixing and microfluidic mixing. The resulting RNA-LNP were first characterized physicochemically, followed by evaluation of their biological performance in vitro and in vivo.
Experimental Model Conditions
The comparative study was performed using identical LNP benchmark formulation conditions for both hand mixing and microfluidic mixing to ensure that observed differences were solely attributable to the LNP mixing strategy.
- Lipid formulation: ALC-0315–based lipid nanoparticle formulation corresponding to the Pfizer/BioNTech mRNA vaccine composition, comprising ALC-0315 (ionizable lipid) , DSPC, cholesterol, and DMG-PEG2000 at a molar ratio of 50:38.5:10:1.5
- Cargo: Yeast RNA (mRNA concentration 0.3 mg/mL)
- Solvent system: Lipids dissolved in ethanol at a total lipid concentration
- Flow rate ratio (FRR): 3:1 (aqueous phase to ethanolic phase) for both mixing approaches
- N/P ratio: 5:1
- Post-processing: Formulated RNA-LNPs were diluted to 10% (v/v) ethanol in water to quench the LNP formulation to achieve a an mRNA concentration of 0.03 mg/mL. Once stabilize RNA-LNP samples were dialysed against 10 mM HEPES buffer (pH 7.4) for 4 hours. LNPs were subsequently reconcentrated to a final lipid concentration of 10–18 mg/mL using Amicon centrifugal filters.
- Experimental design: All formulations were prepared in triplicate to enable robust comparison and assessment of reproducibility
Formulation protocol:
LNP Hand Mixing Protocol
Hand mixing was performed using an optimized manual procedure. Following addition of the ethanolic lipid phase to the aqueous RNA solution, fifty rapid aspiration–dispense cycles were immediately applied using a pipette to promote bulk mixing and nanoparticle formation.
Microfluidic Mixing Protocol
Microfluidic mixing was performed using TAMARA, the advanced microfluidic formulation platform developed by Inside Therapeutics, equipped with a herringbone micromixer to ensure rapid and homogeneous mixing under controlled flow conditions.
Physicochemical characterization of the LNP
Faster mixing leading to smaller nanoparticles
A clear reduction in particle size was observed when using microfluidic mixing compared to hand mixing. Microfluidic formulation yielded LNPs with an average diameter of 82.5 ± 1.0 nm, whereas hand mixing produced larger particles at 102 ± 1.5 nm, corresponding to an approximate 20–30 nm LNP size reduction.
This difference is a direct consequence of the rapid and homogeneous solvent exchange achieved under microfluidic conditions. Faster mixing limits the time window available for nanoparticle growth, leading to earlier stabilization of smaller LNPs sizes. In contrast, the slower and poorly defined mixing kinetics associated with hand mixing allow continued particle growth before stabilization.
Both mixing approaches produced comparable polydispersity indices (PDI), indicating reasonably narrow size distributions in each case.
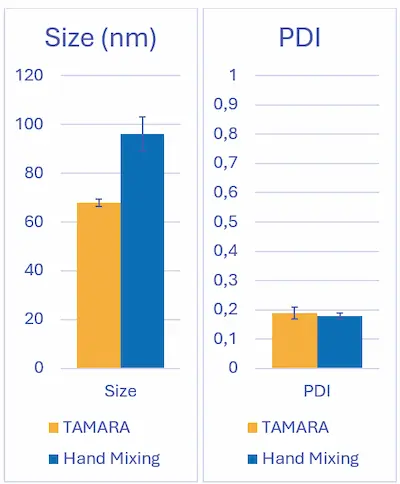
Note that to fully explore the formulation space enabled by microfluidic mixing, additional experiments varying the total flow rate (TFR) would be required. Adjusting TFR directly modulates mixing speed in microfluidic systems and allows systematic tuning of LNP size over a broad range.
Such size control is extremely challenging, if not impossible, with hand mixing, as mixing time and energy cannot be precisely or reproducibly controlled. Similarly, a dedicated operator-dependency study could further highlight the intrinsic variability of hand mixing—driven by factors such as pipetting speed and injection angle—compared to the highly reproducible conditions achieved with microfluidic mixing.
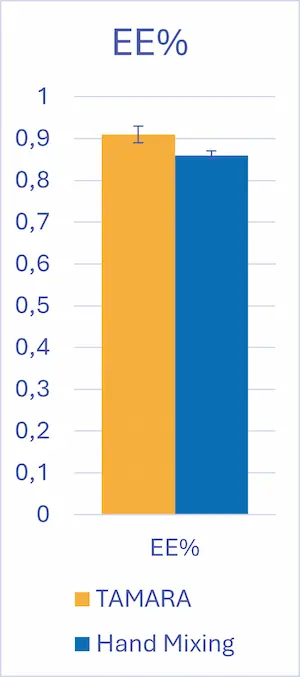
Encapsulation Efficiency
Encapsulation efficiency (EE) remained high for both approaches, with values above 85% and no statistically significant difference between hand mixing and microfluidic mixing.
As a reminder, encapsulation efficiency corresponds to the ratio of encapsulated RNA over the total RNA amount in the final sample. It should not be confused with the Encapsulation Yield (EY) – the ratio of encapsulated RNA over the inital amount of RNA used to formulate the sample. With TAMARA and pipet mixing the yield is >90%, but those values are generally much lower for other formulation systems.
Biological Performance: From In Vitro to In Vivo
In Vitro Expression
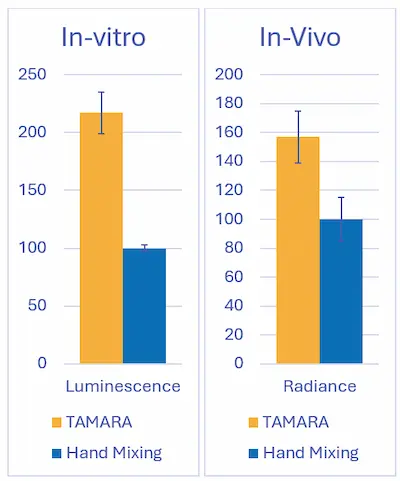
The biological impact of the two mixing strategies was first evaluated using an in vitro transfection assay. HEK293 cells were seeded at 50,000 cells per well and incubated for 24 hours with mRNA-loaded LNPs formulated either by hand mixing or by microfluidic mixing.
Reporter gene expression was quantified by luminescence measurements. Relative luminescence values revealed a clear advantage for microfluidic mixing, with RNA-LNPs achieving more than a two-fold increase in expression compared to hand-mixed formulations. These results demonstrate that improved control over the LNP formation process directly enhances intracellular RNA delivery and functional expression.
In Vivo Radiance
In vivo performance was assessed using bioluminescence imaging following systemic administration of RNA-LNPs. Experiments were conducted in a BALB/c mouse model, with animals receiving an intravenous dose of 0.2 mg RNA/kg.
Consistent with in vitro findings, microfluidic LNPs generated significantly higher radiance signals than hand-mixed LNPs, confirming superior RNA delivery and expression in vivo.
Conclusion on Hand mixing vs microfluidics for LNP formulation
This study demonstrates that the mixing technique used to prepare RNA-LNPs leaves a significant and persistent fingerprint on their critical quality attributes (CQAs). These differences extend beyond standard physicochemical metrics and translate into substantial variations in biological performance, both in vitro and in vivo.
Importantly, this happened as both hand mixing and microfluidic mixing achieved high encapsulation efficiency and comparable PDI values, confirming that the superior biological performance observed with microfluidic mixing is not driven by RNA loading efficiency. Instead, it arises from more subtle yet critical differences in nanoparticle size, internal structure, and population homogeneity introduced during the mixing step.
This distinction becomes increasingly important as the field moves toward next-generation LNPs, where therapeutic performance relies on highly specific CQAs to control biodistribution, cellular uptake, and intracellular processing. In such applications, even minor variations in lipid organization or surface presentation—often invisible to population-averaged characterization—can have profound biological consequences.
These results demonstrate that LNP hand mixing, while convenient for early research, introduces uncontrolled variability in critical quality attributes that directly impacts RNA-LNP biological performance and translational relevance. Findings derived from such non-scalable RNA LNP manufacturing methods may fail to translate when formulations are reproduced using more controlled or industrially relevant processes.
More generally, as the field progresses, LNP formulation must place greater emphasis on well-defined, reproducible, and scalable mixing strategies, and studies should consider and report mixing conditions with the same rigor as lipid composition or biological assays. Only by doing so can results be considered robust, generalizable, and truly translatable.

Looking to improve your LNP formulation using microfluidics?
Reach out to us to learn how we can help!
References
[1] T. Bethiana, A. Aljabbari, Y. Li, H. Mitra, M. Baghbanbashi, G. Harris, S.R. Dasaro, F. Masoomi, F. S. Vago, S. L. Hartzler, M. Figueiredo, L. A. Metskas, P. Vlachos, A. Ardekani, Y. Yeo, K. Ristroph bioRxiv 2025.11.07.687311; doi: https://doi.org/10.1101/2025.11.07.687311







