mRNA
Why is mRNA a breakthrough therapeutic platform?
Messenger RNA (mRNA) has emerged as a powerful therapeutic modality, enabling transient expression of virtually any protein of interest within cells. Its non-integrating nature, rapid development timelines, and broad applicability have positioned mRNA as a versatile platform for vaccines, protein replacement therapies, immuno-oncology, and gene editing. The clinical success of COVID-19 mRNA vaccines has validated this approach and accelerated the development of mRNA-based therapeutics across multiple disease areas.
What is mRNA and how does it work?
mRNA structure
mRNA is a single-stranded nucleic acid molecule that encodes instructions for producing a specific protein. It typically consists of a 5′ cap, a 5′ untranslated region (UTR), a coding sequence, as well as a 3′ UTR, and a 3′ poly(A) tail. These elements play a critical role in stability, translation efficiency, and protein expression levels.
mRNA mechanism of action
Once delivered into the cytoplasm, mRNA is translated by ribosomes to produce the encoded protein. Unlike DNA-based approaches, mRNA does not enter the nucleus or integrate into the genome, resulting in transient and controllable protein expression. This makes mRNA particularly well suited for applications such as vaccination, temporary protein supplementation, and immune modulation.
Where is mRNA applied?
The ability of mRNA to drive transient protein expression makes it a versatile platform across research and therapeutic applications:
- Vaccines: mRNA drives intracellular antigen expression to elicit robust immune responses, as demonstrated by the success of COVID-19 vaccines. mRNA vaccines are now being explored for other viral infections such as influenza, Zika, and rabies, as well as for bacterial infections like tuberculosis, and for personalized cancer vaccines.
- Cancer immunotherapy: mRNA delivers therapeutic proteins — such as cytokines or immune-modulating factors — to stimulate anti-tumor immune responses.
- Gene and genome editing: mRNA can be used to deliver gene-editing components — such as CRISPR-associated (Cas) proteins, base editors, or other modulators — to correct or modulate disease-causing genes. Transient mRNA expression allows a controllable approach for treating genetic and rare disorders while enabling permanent correction of the target gene.
What are the challenges in mRNA therapeutics?
mRNA performance is influenced by both molecular design and delivery strategy.
mRNA design considerations
Effective mRNA therapeutics must overcome design challenges, including poor stability, suboptimal translation, and immune activation. Sequence optimization, such as careful selection of codons and untranslated regions (UTRs), can enhance translation efficiency and protein expression. In addition, chemical modifications — for example, incorporation of modified nucleosides like pseudouridine — are widely used to reduce immunogenicity, improve biological stability, and maximize translational capacity.
mRNA delivery challenges
At the same time, effective delivery is critical. Free mRNA presents significant delivery challenges: it is inherently unstable, highly susceptible to degradation by ubiquitous RNases, and its large size and negative charge prevent passive cellular uptake. Additionally, unmodified mRNA can trigger innate immune responses, potentially limiting efficacy or tolerability. As a result, successful mRNA therapeutics rely on efficient delivery systems capable of protecting the payload, facilitating cellular uptake, and enabling cytosolic release while maintaining an acceptable safety profile.
How to optimize mRNA delivery into cells?
To overcome the previously mentioned delivery barriers regarding free mRNA, scientists have developed a variety of delivery systems, including physical methods (e.g., electroporation) and non-viral nanocarriers (e.g., lipid-based and polymer-based nanoparticles).
Lipid Nanoparticles (LNPs): The gold standard in mRNA therapeutics
Among these approaches, LNPs have emerged as the most clinically advanced platform for mRNA delivery. LNPs are highly customizable, allowing fine-tuning of lipid composition, size, and surface properties. They support targeted delivery, exhibit low cytotoxicity, and are suitable for both in vivo and ex vivo applications. LNPs can be produced at scale and have been validated in clinical settings, most notably in the rapid deployment of COVID-19 mRNA vaccines.
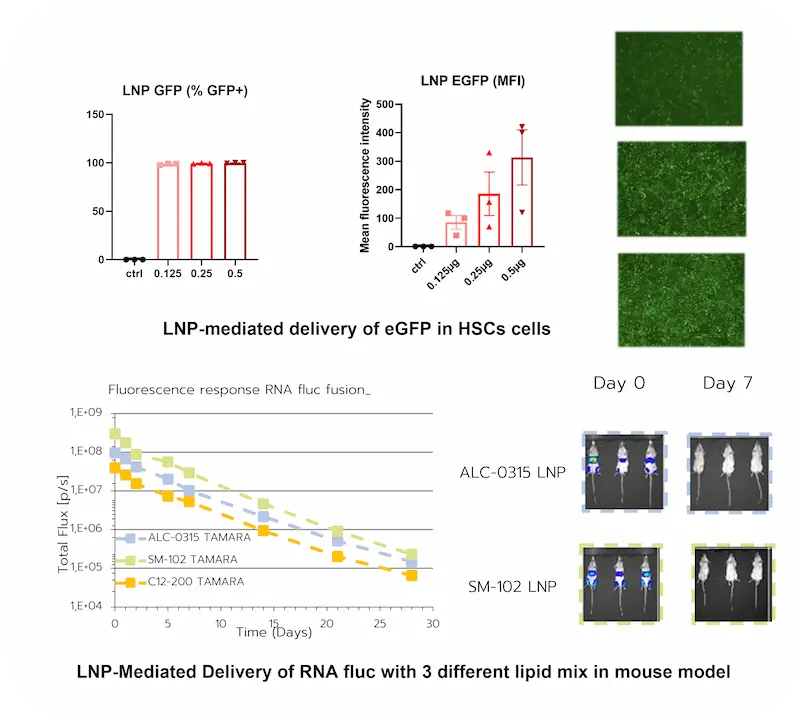
Assessing mRNA-LNP efficiency in vitro & in-vivo
Using reporter genes, mRNA-LNP performance can be evaluated through direct measurement of protein expression in relevant biological models.
mRNA-LNP In vitro (HSC cells):
Using eGFP as a reporter, mRNA-LNP performance was assessed in HSC cells through direct visualization of protein expression after transfection. The results showed:
- Transfection efficiency reaching 100%
- A clear dose-dependent response, demonstrating tunable delivery performance
mRNA-LNP In vivo (mouse):
Complementary in vivo studies can be performed in mice using luciferase as a reporter and three different LNP types. These results showed:
- A strong dependence on the ionizable lipid used, highlighting the critical role of formulation composition
- A progressive decrease in expression over time, with signal dropping by approximately 10-fold per week, consistent with the transient nature of mRNA expression
Together, these results illustrate the efficient of LNP for mRNA delivery across both in vitro and in vivo settings.
Take a deeper dive into the world of nanomedicine
Check our related content and discover how we can help
How we can help
Want to bring mRNA-LNPs into your own lab?
We provide the tools, resources, and expertise you need to work independently with RNA-LNPs:
- TAMARA – our integrated microfluidics platform that empowers you to formulate mRNA-LNPs quickly, efficiently and reliably, producing reproducible nanoparticles with ease.
- Lipid kits for reliable, reproducible & benchmarked RNA-LNP preparation.
- Comprehensive resources, including RNA-LNP protocols, application notes, and best practices to support your workflow from design to formulation.
- Training programs that teach practical formulation and handling skills.


Looking to run proof-of-concept RNA-LNP studies?
Our tailored formulation services help you quickly evaluate mRNA-LNPs in your own lab or to accelerate your experimental progress:
- LNP design, based on either trusted benchmark compositions (Moderna, Pfizer…) or custom formulations.
- Formulation using microfluidics & purification to ensure a controlled, reproducible and efficient development process.
- Comprehensive CQA characterization, including size, PDI, and encapsulation efficiency, with advanced options such as morphology and payload loading.
- Lipids and/or reporter mRNA supply for an end-to-end project solution.