Lipid Nanoparticles (LNPs)
How are LNPs revolutionizing RNA delivery?
Lipid nanoparticles (LNPs) represent the most advanced and clinically validated platform for RNA delivery to date. They protect any nucleic acids (mRNA, siRNA, saRNA, CircRNA…) from enzymatic degradation, facilitate cellular uptake, and enable endosomal escape — key factors that determine therapeutic efficacy. Having been validated clinically through mRNA COVID-19 vaccines, LNPs are now central to RNA-based therapeutics in infectious diseases, oncology, and gene editing. Their modularity and tunability make them invaluable for preclinical research and the subsequent translation to clinical studies.
What’s behind an LNP?
LNP composition & structure
LNPs are typically composed of four lipid constituents:
- Ionizable lipids (40 – 50 mol%) – Neutral at physiological pH but positively charged in acidic environments, promoting RNA encapsulation during formulation and endosomal escape in target cells. e.g., DLin-MC3-DMA, SM-102, ALC-0315, LP-01, C12-200.
- Helper phospholipids (10 – 15 mol%) – Provide structural integrity to the lipid bilayer. They can improve intracellular delivery efficiency or extend particle’s circulation time. e.g., DSPC, DOPE.
- Cholesterol (38 – 50 mol%) – Enhances lipid membrane stability, reduces interaction with serum proteins, modulates membrane fluidity, and facilitates fusion with cellular membranes.
- PEG-lipids (1.5 – 2 mol%) – Minimize aggregation and opsonization, improve stability and circulation time. e.g., PEG-c-DMG, PEG-DMG, ALC-0159.
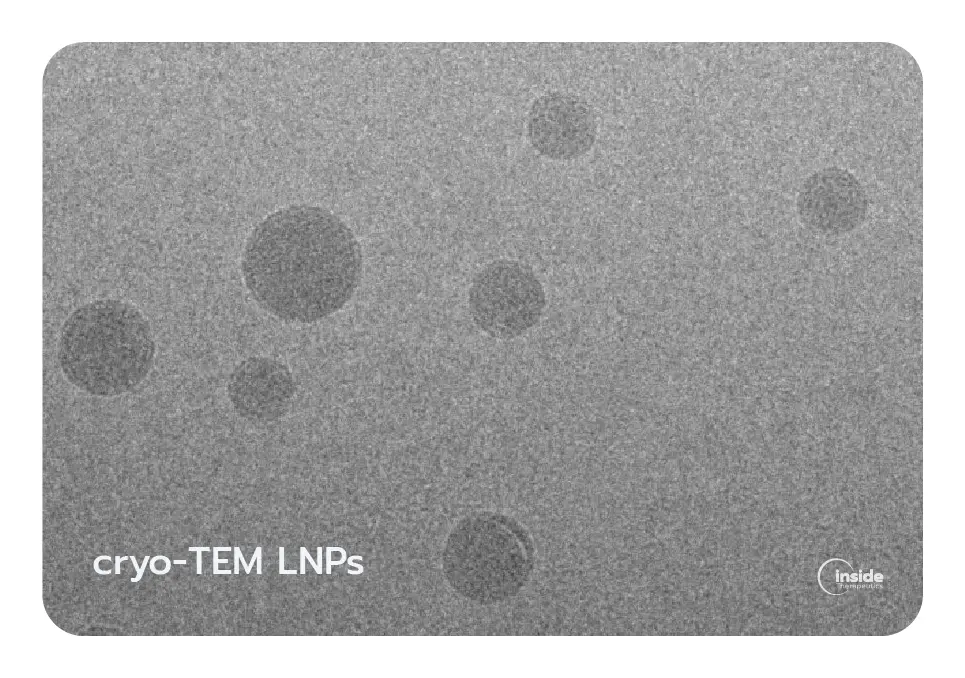
The combination of these lipids produces nanoparticles generally in the 50–200 nm range with high encapsulation efficiency (>90% in optimized formulations). LNPs can be formulated to carry various RNA payloads, including mRNA, siRNA, and CRISPR components, and are widely applied in vaccines, protein replacement therapies, cancer immunotherapy, and gene editing. Cryo-electron microscopy studies have shown that LNPs can exhibit distinct internal morphologies, such as solid-core, biphasic, or multiphasic structures, depending on lipid composition and formulation parameters.
How do LNPs deliver RNA into cells?
Cellular uptake of LNPs
LNPs are primarily internalized by cells via endocytosis, which includes phagocytosis (takes place specifically in professional phagocytes such as macrophages, monocytes, and neutrophils) and pinocytosis (occurs in nearly all cell types). Among multiple mechanisms of pinocytosis, clathrin-mediated endocytosis is the main route that nucleic acid-loaded LNPs are taken up by cells.
Intracellular trafficking & endosomal escape
Following internalization, LNPs are trafficked through the endolysosomal system, where vesicles progressively acidify as they mature from early endosomes (pH 6.0-6.5) to late endosomes (pH 5.0-5.5) and lysosomes (pH 4.5-5.0). This acidic environment activates the ionizable lipids within the nanoparticle, which transition from a neutral to a positively charged state when the pH value becomes lower than their acid dissociation constant (pKa). The resulting electrostatic interactions with negatively charged endosomal membrane lipids induce membrane destabilization, allowing the RNA cargo to escape into the cytoplasm, where it can exert its biological function. Without successful endosomal escape, RNA payload is ultimately degraded in hydrolase-rich environment of lysosomes or recycled back to the membrane, limiting therapeutic activity. Efficient endosomal escape is therefore a key determinant of LNP-mediated RNA delivery.
What makes LNPs so effective — and what still needs to be optimized?
Key advantages of LNPs
- Modular and tunable design adaptable to multiple RNA payloads
- Potential for tissue targeting through surface functionalization
- Favorable biocompatibility and low cytotoxicity
- Suitable for both in vivo and ex vivo applications
- Scalable manufacturing
- Clinical validation
Active areas of research
- Endosomal escape efficiency: Only ~1–4% of LNP-delivered RNA currently reaches the cytosol. Novel ionizable lipid chemistries and membrane-destabilizing strategies are being explored to close this gap.
- Tissue-specific targeting: Systemically administered LNPs naturally accumulate in the liver. Redirecting delivery to other organs — lung, spleen, brain, tumors — or cells – HSCs, CAR-T… – is one of the most active frontiers in the field.
Current approach generally involve passive strategies (lipid composition tuning, SORT lipids), active targeting (antibody-conjugated LNPs, nanobodies, bispecific ligands) or hybrid structures (LNP-EVs) - Immune profile optimization: PEGylation can influence innate immune activation. Ongoing work focuses on balancing stealth properties with controlled adjuvant effects — a critical factor for both vaccines and therapeutic applications.
Why do LNP characteristics matter?
Optimizing LNP formulations
LNPs should be carefully optimized to enhance stability and maximize RNA delivery to the target site while minimizing toxicity. This requires tuning critical process parameters (CPPs) — including lipid composition and ratios, buffer conditions, formulation method and parameters, and downstream processes — to achieve the desired critical quality attributes (CQAs) of the nanoparticle, such as particle size, polydispersity index (PDI), zeta potential, and morphology.
Key LNP attributes
- Particle size and polydispersity index (PDI): Dictates cellular uptake and biodistribution profiles.
- Surface charge (ζ-potential): Impacts interaction with serum proteins, stability, and toxicity.
- Encapsulation efficiency: Determines effective dose and minimizes RNA waste.
- Morphology: Affects cellular uptake, payload release, stability, and biodistribution.
How are LNPs prepared?
Effect of formulation methods on LNP quality
How LNPs are produced plays a decisive role in defining their critical quality attributes (CQAs), including particle size, size distribution, surface charge, morphology, encapsulation efficiency, and overall yield. These properties ultimately determine biological behavior such as circulation time, cellular uptake, biodistribution, toxicity, and therapeutic outcome. Even minor variations in processing conditions can result in meaningful differences in nanoparticle performance, highlighting the importance of tightly controlled and reproducible manufacturing techniques.
LNP formulation approaches
Early bulk techniques, such as thin-film hydration or ethanol injection, provide straightforward and accessible entry points for LNP preparation. As the field has matured, more advanced approaches have been adopted, including microfluidic mixing and impingement jet mixing (IJM). Microfluidic systems are particularly well suited for rapid formulation screening and reproducible preclinical development, whereas IJM platforms support the higher throughput required for clinical manufacturing. Each approach offers distinct advantages and limitations with respect to scalability, and batch-to-batch consistency, and operational complexity. Choosing the most appropriate formulation strategy ultimately depends on the development stage and the intended application.
Why use microfluidics for RNA–LNP formulation?
Controlled LNP self-assembly
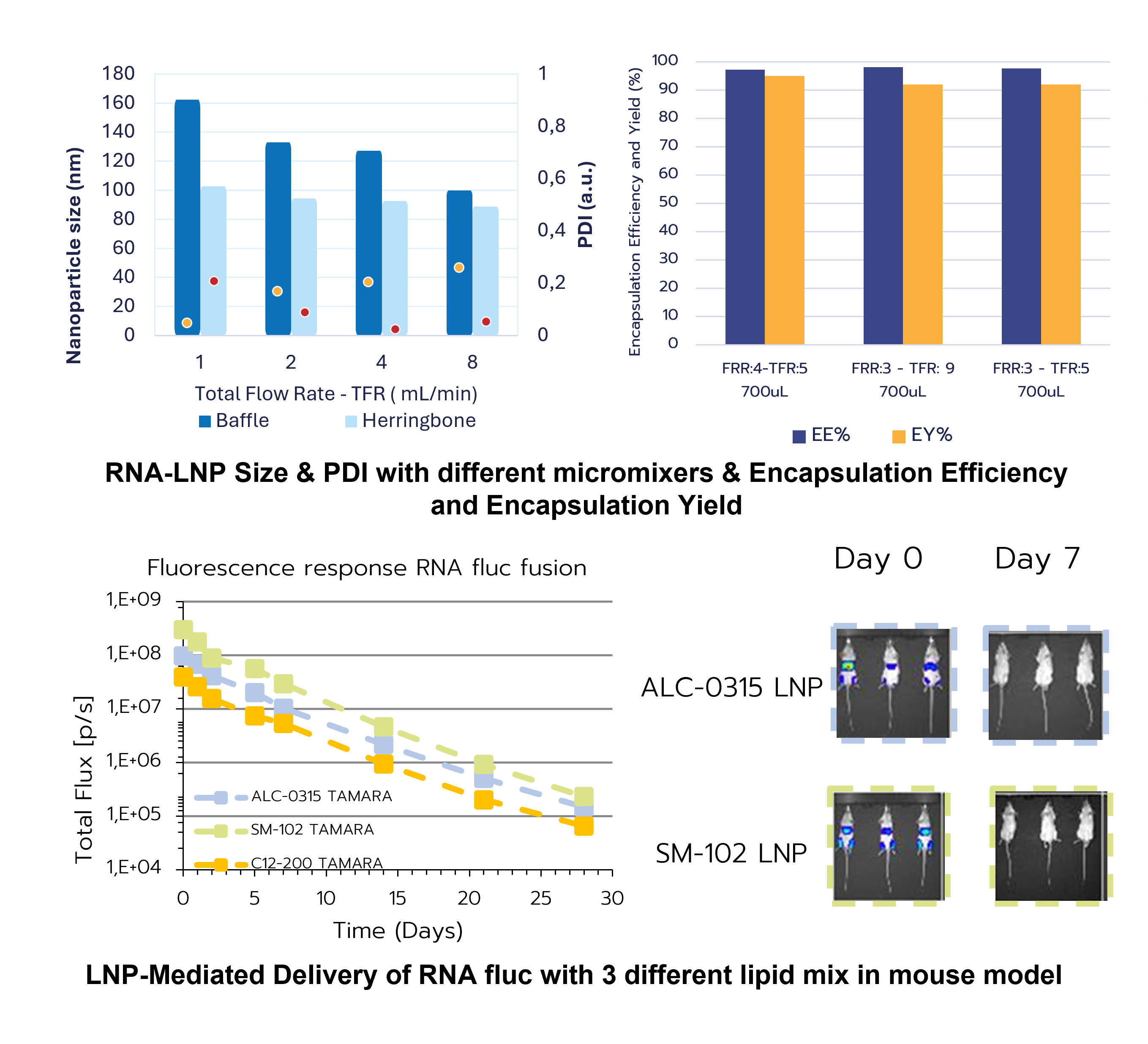
Microfluidic systems allow highly reproducible RNA–LNP production by precisely controlling the mixing of lipids (dissolved in an organic solvent, typically ethanol) and aqueous RNA solutions at the microscale. Rapid solvent exchange drives spontaneous nanoparticle self-assembly, producing uniform particles with narrow size distributions. By adjusting parameters such as the flow rate ratio (FRR) between aqueous and organic phases and the total flow rate (TFR), key properties — including particle size, PDI, and encapsulation efficiency — can be finely tuned. Modern microfluidic platforms incorporate various micromixer designs (e.g., T- or Y-junctions, hydrodynamic flow focusing, staggered herringbone, baffle, and toroidal mixers), each offering distinct benefits and trade-offs.
Accelerated & resource-efficient LNP development
Because microfluidics operates at low volumes with excellent batch-to-batch consistency, it supports rapid screening and optimization of formulations while minimizing reagent consumption. This makes it a powerful preclinical platform for systematically refining RNA–LNP systems and accelerating their path toward in vivo evaluation and clinical translation.
Take a deeper dive into the world of nanomedicine
Check our related content and discover how we can help
How we can help
TAMARA
RNA-LNP Formulation System
Fast & User-friendly operation
Rapid RNA–LNP formulation with an intuitive workflow
Integrated R&D platform
Flexible working range (0.2-30 mL; 1 µg-5 mg of RNA per run)
Efficient use of materials
Reusable chips & zero formulation losses
Precision & Reproducibility
Microfluidic efficiency & precision


Lipid Kits
LNP Starter Kits – Premix of Lipids to get started easily.
Simplified start to LNP formulation
Reduced preparation complexity
Trusted formulation reference
Composition based on LNPs used in authorized mRNA vaccines
Practical scale for R&D workflows
100 mg total lipids/kit (≈50 mL RNA–LNP formulation), enabling multiple formulation runs
High-quality lipid materials
GMP-like lipid components for reliable nanoparticle performance
Ready to Get Started with RNA-LNPs?
Explore our Services & Trainings
CRO formulation
We formulate, purify, characterize, and optimize your RNA-LNPs — from proof of concept to preclinical batches.
Hands-on Training
1 to 3 days at our lab or yours. LNP fundamentals through formulation with TAMARA. You leave with protocols, data, and autonomy.
Let us know what you need. We handle the rest.