
Abstract
This introduction to solid lipid nanoparticles (SLNs) highlights their role as biocompatible, versatile delivery systems that enhance drug stability, bioavailability, and controlled release. It explains core components, solid lipids, surfactants and discusses about scalable fabrication methods and multi-route applications.
Over the last few decades, the significance of lipid-based nanoparticles (LBNs) development has greatly increased in the realm of biomedicine. Among these LBNs, solid lipid nanoparticles (SLNs) are part of the most promising ones, due to their unique properties. Their development started to overcome some limitations met with traditional nanocarriers like liposomes, such as physicochemical stability or poor encapsulation efficiency. SNLs benefit from unique properties such as large surface area, low toxicity, and can encapsulate both lipophilic and hydrophilic drugs. These carriers are composed of a crystalline lipid core stabilized by a surfactant and co-surfactant shell and their encapsulation properties differ depending on the nature of lipids and surfactants used. Lipophilic drugs are encapsulated between fatty acid chains, between the lipid layers and inside crystal imperfections, while low doses of hydrophilic drugs can be incorporated in the lipid core. Modifying these carriers’ composition makes them tailorable to specific applications, like controlled release mechanism for drug delivery or RNA encapsulation.
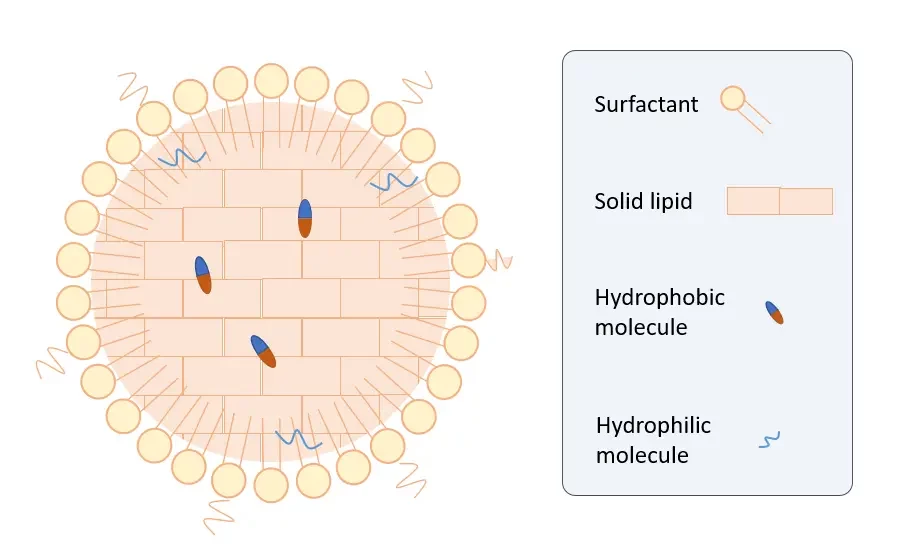
Solid lipid nanoparticle formulation
Despite the structural differences between SLNs and other lipid-based nanoparticles, the mechanism responsible for their formation is similar.
A large variety of lipids can be used to formulate SLNs: switching a lipid for another one or changing the relative proportion of the components changes the crystal structure of the lipid matrix. Mono-, di-, and triglycerides, fatty acids, steroids, and waxes are the lipids that are usually used when it comes to formulating SLNs, due to their low toxicity and their melting point above the body temperature, to maintain the solid core structure in vivo.
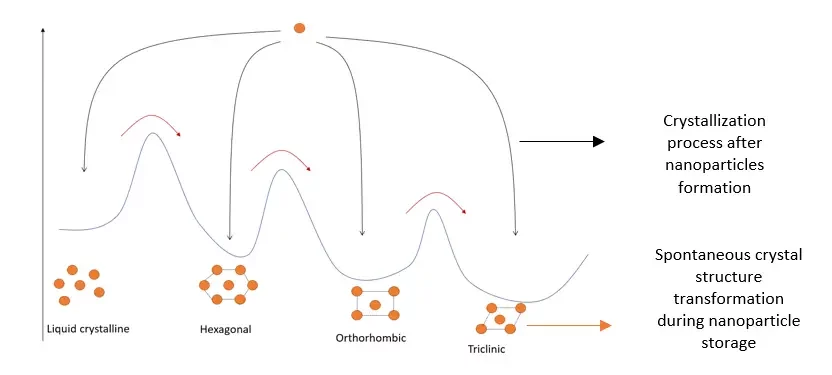
A crucial parameter of lipid solidification that requires specific attention is the possible polymorphic transformation that can occur after the SLN production. By being able to crystallize in different possible crystal species during their production, the SLNs can then go through polymorphic transitions to reassemble into more stable structures, leading to possible drug ejection and lowering drug entrapment efficiency. A higher-ordered crystal lattice leads to fewer imperfections, thus leading to a lower drug encapsulation capacity.
The selection of adequate surfactants arises from the lipid since physicochemical compatibility is required between them. A combination of surfactants or the addition of co-surfactants is usually used to prevent particle coalescence. Also, this choice depends on the administration route, either parenteral or oral. Amphiphilic molecules such as monoglycerides of long-chain fatty acids, or phospholipids can be used as surfactants.
Solid lipid nanoparticles production methods
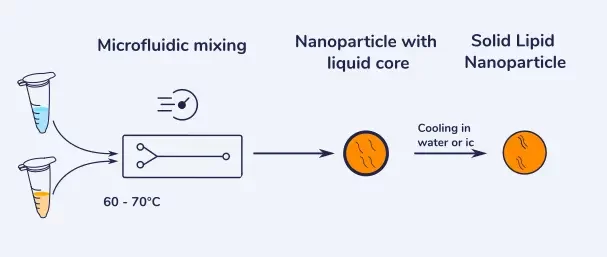
Different methods On the other hand microfluidics allows for the optimal control of the nanoparticle’s final characteristics, and thus an optimized delivery, but considering the use of solvent, an extra step of solvent extraction is required.
Use of solid lipid nanoparticles
LBNs started being developed to overcome important issues in the biomedical realm, especially for cancer treatment. Due to their high toxicity and their low biodistribution, molecules commonly used for chemotherapy need encapsulation to increase their half-life in the body. Moreover, the development of these nanocarriers enables specific cells targeting with PEG-coated and ligand-coated LBNs, leading to reduced toxicity for healthy cells.
Among the different LBNs, SLNs are part of the most promising ones for anticancer drug encapsulation. SLNs’ lipophilic structure increases permeability through the intestinal wall, allowing possible oral administration. Also, SLNs’ enhanced penetration through the skin has also been studied, making them possible carriers for drug delivery to melanomas directly through the skin. Coating SLNs with PEG and specific antibodies further enhances these targeting properties.
In addition to their tumor-targeting properties and the introduction of new administration routes for cancer treatment, the use of SLNs for delivering a wide variety of drugs is being studied, due to their drug release mechanism. The drug present on the SLNs’ surface shows an immediate release effect, while the drug encapsulated in the lipid core won’t be released until the core is degraded, leading to tailorable release mechanisms by making the lipid core sensible to temperature, pH, etc, which makes SLNs suitable for macromolecules delivery, like mRNA, vitamins or antimicrobial.
Advantages of SLN
SLNs stand out from the other LNPs for different reasons. First, as already mentioned, due to their structure, SLNs can encapsulate both hydrophilic and lipophilic drugs, but they also improve the stability of the pharmaceuticals they carry. By improving the circulation time of the drug in the body and protecting it from degradation better than other LBNPs, they enhance the drugs’ biodistribution, as well as allow a targeted and controlled drug release. Moreover, since most lipids used while formulating SLNs are biodegradable, SLNs benefit from low toxicity/ high biocompatibility. This high biocompatibility is also enhanced when SLNs are formulated with microemulsion methods. In this regard, economical large-scale production can easily be achieved for SLNs. Finally, SLNs encapsulate a wide variety of molecules, from antitumoral drugs to ARN or cosmetical active principle, and can be administered through different routes thanks to their stability, such as oral, parenteral, nasal, or ocular.
Disadvantages of SLN
Despite these advantages, SLNs also suffer from a few drawbacks. First, the necessary use of surfactant to synthetize SLNs can induce toxicity, regarding the surfactants’ nature and concentration. Also, due to their highly crystallized structure, SLNs lack encapsulation volume, leading to poor drug loading capacity, combined with potential drug expulsion during post-production polymeric transitions. SLNs’ drug loading capacity are even lower for hydrophilic drugs; however, formulating SLNs with a double emulsion method can increase this hydrophilic drug loading capacity. The use of complex lipids to increase the number of crystal imperfections can also counter this major drawback. Moreover, a new generation of LNPs, called nanostructured lipid carriers (NLCs), was developed to overcome these limitations, by adding small amounts of liquid lipids into solid lipids, creating general imperfections in the crystal and enhancing drug loading capacity. Lipid drug conjugates (LDCs) were also created to counter the SLNS’ low hydrophilic drug loading capacity by increasing the drug solubility in the lipid melt during the production.
Conclusion on solid lipid nanoparticles
Due to the numerous applications linked to SLNs and their tailorable properties, these nanocarriers’ synthesis require precision, which can be more easily achieved by using microfluidic devices. This method also ensures a better repeatability over the produced SLNs, and the possibility of large-scale production.

Want to learn more about SLN manufacturing methods?
Speak to our application scientist!







