Gene Editing
Can gene editing cure disease at its source?
Advances in gene editing are transforming medicine, turning what was once science fiction into clinical reality. Technologies like CRISPR-Cas allow precise modulation of disease-causing DNA and RNA sequences, offering hope for conditions ranging from inherited disorders to cancer and infectious diseases. Already, these tools have moved beyond the lab, with therapies like Casgevy achieving FDA approval and highly personalized treatments — such as base editing for rare metabolic disorders — demonstrating the potential for patient-specific interventions. As gene editing continues to evolve, it promises not just to manage disease, but to fundamentally rewrite the genetic code behind it.
Targeting the roots of genetic disorders
The burden of genetic diseases
A substantial number of inherited disorders arise from defined mutations within the human genome, leading to dysfunctional or absent proteins that disrupt cellular homeostasis. Conditions such as sickle cell disease, β-thalassemia, and cystic fibrosis impose lifelong clinical burdens, often requiring chronic management rather than curative intervention. Beyond rare monogenic disorders, genetic drivers also play roles in cancer progression and infectious diseases.
Despite decades of therapeutic innovation, most treatments remain symptomatic or palliative. They address downstream consequences rather than correcting the underlying molecular defect. This creates an urgent need for strategies capable of precise, durable modification of disease-causing genetic sequences.
From disease management to molecular correction
Gene editing introduces the possibility of repairing, silencing, or rewriting pathogenic DNA sequences directly within patient cells. Rather than compensating for defective proteins, genome engineering aims to restore endogenous gene function or permanently disrupt harmful pathways. This shift — from disease management to genetic correction — marks a paradigm change in therapeutic medicine.
What gene editing technologies are available?
Gene editing technologies & the rise of CRISPR-Cas
Several gene editing platforms have been developed to modify DNA, including Zinc-Finger Nucleases (ZFNs) and Transcription Activator-Like Effector Nucleases (TALENs). These systems rely on engineered proteins to recognize specific DNA sequences, which makes them technically demanding, time-intensive, and less flexible for targeting multiple genes simultaneously. In contrast, CRISPR-Cas provides a highly adaptable and programmable approach, where a short RNA sequence guides a nuclease, such as Cas9, to precise genomic loci. This RNA-guided system allows rapid re-targeting to different genes, greater scalability, and broader applicability across diverse disease contexts.
CRISPR-Cas technology offers a versatile platform for therapeutic manipulation of both the genome and transcriptome. Cas9 and Cas12 nucleases create targeted double-strand DNA breaks, allowing gene disruption or correction via the cell’s natural repair pathways. Base editors allow precise single-nucleotide modifications, while prime editors enable accurate insertions without causing double-strand breaks. Cas13 targets RNA transcripts, providing a means for post-transcriptional regulation. In addition, epigenome editing can modulate gene expression, either activating or repressing specific genes. Overall, these mechanisms provide diverse strategies for correcting genetic disorders, reprogramming cells, and intervening in disease pathways.
Where is gene editing being applied?
CRISPR-based gene editing has rapidly emerged as a transformative tool for developing therapies across a wide range of diseases. By enabling precise modification of DNA and RNA, CRISPR allows researchers and clinicians to repair disease-causing mutations, engineer specialized cells, fight infectious agents, and many more.
Key therapeutic applications
- Genetic disorders – CRISPR can repair mutations in genes responsible for inherited disorders such as sickle cell disease, β-thalassemia, Cystic Fibrosis (CF), Duchenne Muscular Dystrophy (DMD), and severe combined immunodeficiency (SCID).
- Oncology & immune engineering – CRISPR is used to disrupt oncogenes or correct mutations in tumor suppressor genes, enabling targeted interventions. It has significantly advanced immune cell therapies, including CAR-T and CAR-NK cell engineering, by enhancing tumor recognition, boosting cytotoxicity, and overcoming inhibitory checkpoints. Additionally, CRISPR helps modulate the tumor microenvironment (TME) to improve treatment outcomes. It also enables the design of oncolytic viruses that selectively infect and kill cancer cells while sparing healthy tissue.
- Infectious diseases – CRISPR systems can selectively target viral DNA or RNA and antimicrobial resistance genes in pathogens, offering potential treatments for viral infections such as HIV-1, HBV, HPV, SARS-CoV-2, and multidrug-resistant bacterial infections.
How does CRISPR-based gene editing translate to clinics?
From ex vivo success to in vivo precision
The therapeutic potential of CRISPR became a reality with the approval of Casgevy, developed by Vertex Pharmaceuticals and CRISPR Therapeutics, for treating sickle cell disease and β-thalassemia. This ex vivo therapy delivers Cas9–guide RNA ribonucleoprotein complexes to hematopoietic stem cells (HSCs) via electroporation, disrupting the BCL11A gene and reactivating fetal hemoglobin (HbF) production
To overcome the limitations of ex-vivo applications, which remain complex and extremely costly, CRISPR is now beginning to demonstrate its promise in vivo, where genome editing components are delivered directly into the body.
A striking example is the highly personalized treatment of “Baby KJ,” a patient born with carbamoyl phosphate synthetase 1 (CPS1) deficiency, a rare metabolic disorder characterized by toxic ammonia accumulation. In this case, clinicians administered a CRISPR base editor complexed with a patient-specific guide RNA using lipid nanoparticles (LNPs), enabling precise correction of the single pathogenic nucleotide in vivo. This milestone illustrates the rapid evolution of CRISPR therapies: from standardized ex vivo workflows to mutation-specific, individualized in vivo genome editing, opening the door to treating a broader range of genetic diseases directly at their source.
What delivery technologies enable gene editing?
Overview of gene delivery strategies
Delivering CRISPR components safely and efficiently is a critical determinant of therapeutic success. Gene editing relies on transporting nucleic acids (e.g., mRNA, pDNA, sgRNA) or ribonucleoproteins (RNPs) into target cells to repair mutations, introduce new functions, or modulate gene expression. Delivery faces intrinsic challenges, including degradation by enzymes, poor cellular uptake, and negative charges on nucleic acids that hinder cell membrane crossing. To overcome these barriers, a range of delivery strategies has been developed, broadly classified into viral and non-viral approaches. Viral vectors — such as adenoviruses (AVs), lentiviruses (LVs), and retroviruses (RVs) — harness the natural ability of viruses to transfer genetic material efficiently, achieving high expression levels. However, they carry risks including immune activation, toxicity, insertional mutagenesis, and large-scale manufacturing complexity. Non-viral systems, including physical methods like electroporation, microinjection, and hydrodynamic injection, as well as chemical carriers such as lipid nanoparticles (LNPs), polymer-based nanoparticles, and inorganic nanoparticles, are increasingly investigated considering their safer profiles, with the later also allowing for in-vivo approaches.
Why choose LNPs for CRISPR components delivery?
Among non-viral delivery approaches, LNPs have emerged as a effective carriers both for in vitro, ex vivo, and in vivo CRISPR gene editing. Compared with electroporation, which is largely limited to in vitro or ex vivo applications, and viral vectors, which pose risks of toxicity and genomic integration, LNPs offer a highly customizable, safe, and clinically scalable platform.
LNPs can encapsulate various formats of CRISPR cargo, such as Cas9 mRNA, Cas9 RNP and single guide RNAs (sgRNAs), protecting them from enzymatic degradation and enabling cytosolic delivery via endosomal escape. They are generally biocompatible and biodegradable, reducing potential toxicity, and can be functionalized to improve targeting to specific cell types, enhancing therapeutic efficacy. Taken together, LNPs represent a clinically validated, versatile, and powerful platform for bringing CRISPR therapies to patients.
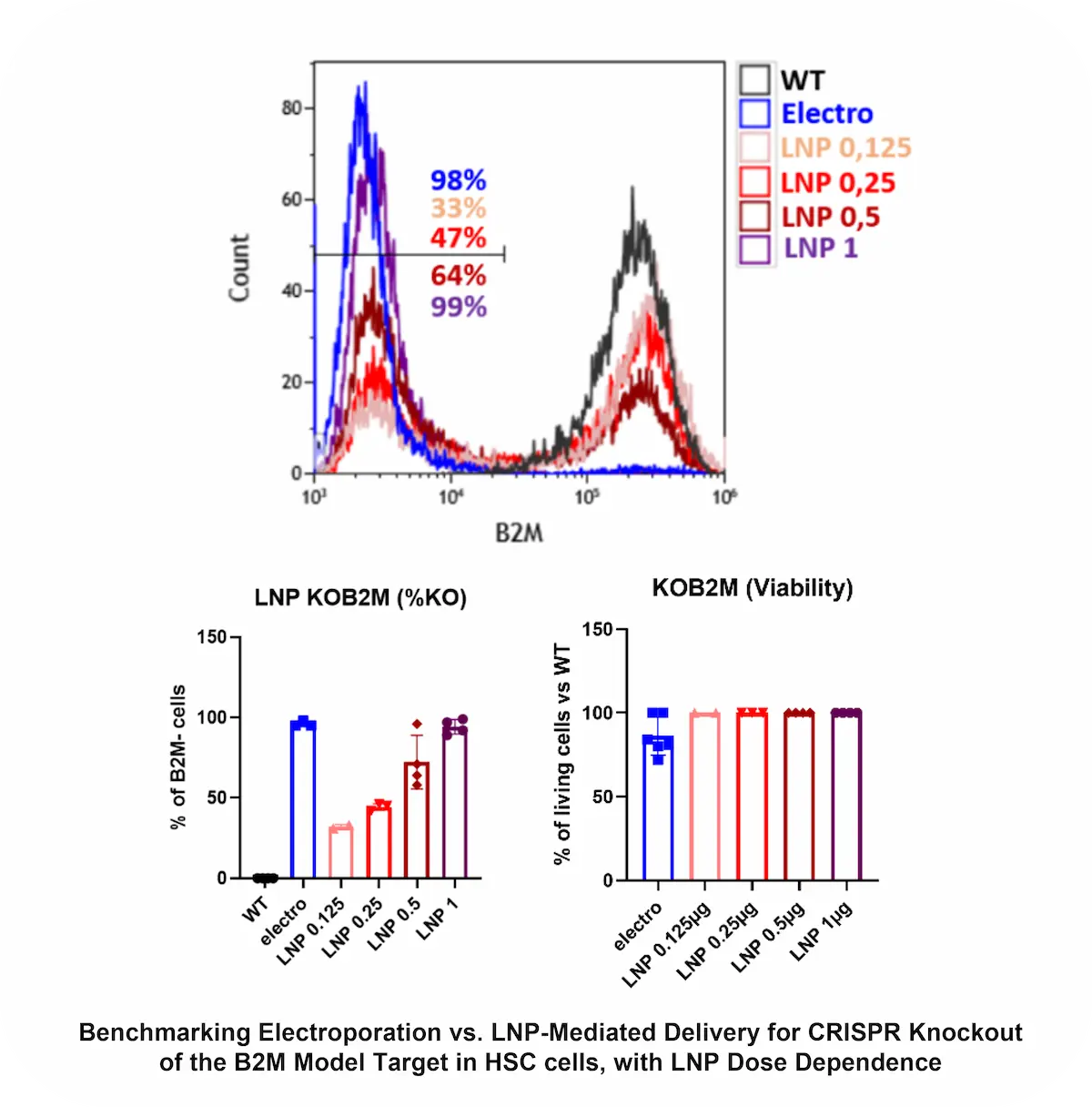
When benchmarked against electroporation in a B2M CRISPR knockout model on HSCs cells, LNPs show that:
- At their optimal doses, both approaches achieved similar knockout efficiencies.
- LNP delivery maintained near zero cytotoxicity, whereas electroporation caused substantial cell death (around 20%).
- Electroporation required a 10 fold higher sgRNA dose for similar results
Proving that LNP Lcan match the performance of conventional ex-vivo transfection methods enabling future in-vivo CRISPR approaches.
Take a deeper dive into the world of nanomedicine
Check our related content and discover how we can help
How we can help
Want to bring RNA-LNPs into your own lab?
We provide the tools, resources, and expertise you need to work independently with RNA-LNPs:
- TAMARA – our integrated microfluidics platform that empowers you to formulate RNA-LNPs quickly, efficiently and reliably, producing reproducible nanoparticles with ease.
- Lipid kits for reliable, reproducible & benchmarked RNA-LNP preparation.
- Comprehensive resources, including RNA-LNP protocols, application notes, and best practices to support your workflow from design to formulation.
- Training programs that teach practical formulation and handling skills.


Looking to run proof-of-concept RNA-LNP studies?
Our tailored formulation services help you quickly evaluate RNA-LNPs in your own lab or to accelerate your experimental progress:
- LNP design, based on either trusted benchmark compositions (Moderna, Pfizer…) or custom formulations.
- Formulation using microfluidics & purification to ensure a controlled, reproducible and efficient development process.
- Comprehensive CQA characterization, including size, PDI, and encapsulation efficiency, with advanced options such as morphology and payload loading.
- Lipids and/or reporter RNA supply for an end-to-end project solution.