Peptide-based Nanoparticles (PBNs)
Can short peptides unlock safe and efficient nucleic acid delivery?
Peptide-based nanoparticles (PBNs) represent a class of non-viral carriers formed from short synthetic or naturally derived peptides, most commonly cell-penetrating peptides (CPPs). Typically composed of up to 30 amino acids, CPPs originate from human, viral, or synthetic sequences and were identified over two decades ago as promising intracellular delivery vectors. PBNs offer low immunogenicity, reduced cytotoxicity, and biocompatibility. Their modular design allows easy chemical modification, making them attractive candidates for nucleic acid delivery across a broad range of therapeutic applications such as gene therapy or next generation cancer therapies.
What are peptide-based nanoparticles?
Amphipathic properties & self-assembly
Early cell-penetrating peptide (CPP)-based delivery systems relied on covalent conjugation between peptides and nucleic acids. Over time, non-covalent approaches became predominant, leading to the development of peptide-based nanoparticles (PBNs). In these drug delivery systems, nanoparticle formation is driven primarily by electrostatic interactions between positively charged amino acids — such as arginine, lysine, and histidine — and the negatively charged phosphate backbone of nucleic acids. However, charge interactions alone are not sufficient to produce stable nanostructures. Efficient self-assembly also requires hydrophobic domains — either arising from intrinsic nonpolar amino acids within the peptide sequence or introduced via the grafting of fatty acid chains, commonly at the N-terminus. This combination of cationic and hydrophobic regions — often organized through amphipathic primary, secondary, or tertiary structures — enables nucleic acid condensation and formation of compact nanoparticles.
The resulting PBNs typically range from 60 to 150 nm in diameter and exhibit a positive surface charge, generally between +10 mV and +40 mV depending on formulation conditions. This surface charge reflects the peptide composition and carrier-to-cargo ratio, and facilitates membrane interaction and cellular uptake.
Peptide families & chemical modifications
CPP-based systems span multiple peptide families, including poly-cationic peptides, GALA/KALA/RALA derivatives, PepFect/NickFect variants, CADY-K/RICK, WRAP, C6, and Mpge peptides. To improve stability, targeting precision, and overall biological performance, these peptides can be further engineered through chemical modifications, including PEGylation, conjugation of targeting ligands, or incorporation of organelle-directing sequences.
How do PBNs deliver nucleic acids into cells?
Cellular entry & intracellular routing
Following administration, PBNs can enter cells either through direct membrane translocation or endocytosis-dependent mechanisms. After cellular uptake, successful delivery requires release of the nucleic acid cargo into the cytosol. Depending on the therapeutic modality, nucleic acids may then: silence gene expression (e.g., siRNA), induce protein translation (e.g., mRNA), modulate splicing (e.g., ASOs), enable gene expression (e.g., pDNA), or support genome editing through CRISPR-based systems. Efficient intracellular trafficking and endosomal escape are therefore central to biological activity.
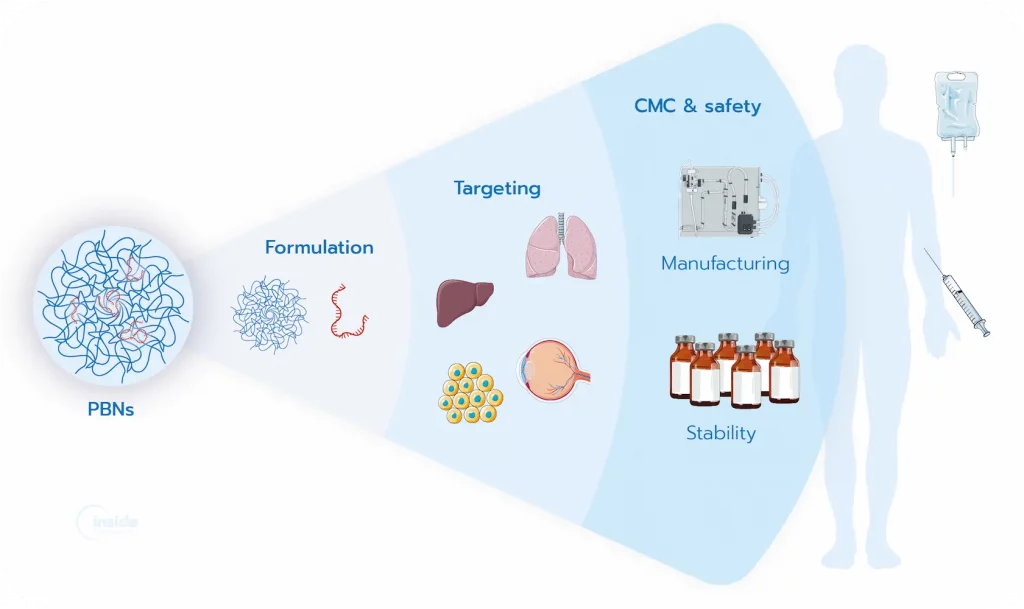
What are the strengths and current challenges of PBNs?
PBNs represent an increasingly attractive non-viral platform for nucleic acid delivery, offering an alternative to viral vectors as well as lipid- and polymer-based systems.
Advantages of peptide-based nanoparticles
- Low cytotoxicity & biodegradability – Peptides degrade into amino acids, supporting safety and tolerability.
- Versatile nucleic acid compatibility – Capable of delivering various nucleic acid formats, including siRNA, mRNA, pDNA, and ASOs.
- Simple synthesis & formulation – Automated peptide synthesis and single-step nanoparticle assembly.
- Modular functionalization – Easy chemical modification can enable targeting, shielding, and stimuli-responsiveness.
- Efficient membrane interaction – Intrinsic cell-penetrating capability enhances intracellular access.
Current limitations & optimization needs
- Short systemic circulation time – Rapid clearance due to size and surface charge.
- Immune clearance – Positive surface charge may enhance opsonization and subsequent clearance by the mononuclear phagocyte system (MPS).
- Need for enhanced targeting specificity – Active targeting strategies are required to improve targeted delivery in specific organelle/cell/tissue.
- Limited clinical translation – Even though there are a substantial number of preclinical studies focused on PBNs, clinical adoption remains limited.
Advanced strategies such as PEGylation, fatty acid grafting, targeting ligands, and stimulus-responsive designs (pH- or enzyme-triggered systems) are being developed to address these limitations.
Why do PBNs characteristics matter?
Optimizing PBN formulations
The biological performance of PBNs is strongly influenced by their physicochemical properties, which are governed by critical process parameters (CPPs). Carefully defined formulation conditions — including peptide-to-cargo ratio, buffer conditions, and mixing parameters — are essential to produce homogeneous and reproducible nanoparticles suitable for both in vitro and in vivo applications.
Key PBN attributes
- Particle size (60–150 nm) – Influences circulation time, cellular uptake, and biodistribution.
- Polydispersity index (PDI < 0.30) – Ensures homogeneity and reproducibility.
- Surface charge (+10 to +40 mV) – Affects membrane interaction, serum stability, and toxicity.
Careful control of these attributes directly impacts therapeutic efficacy and safety.
How does microfluidics enhance PBN production?
Controlled and reproducible PBN assembly
Microfluidic preparation allows tight modulation of process parameters (e.g., mixing conditions, peptide-to-cargo ratios, and buffer conditions), promoting uniform self-assembly of PBNs. This controlled peptide self-assemply approach consistently produces nanoparticles within the desired size range with low polydispersity, ensuring high homogeneity, reliable biological performance, and reproducible batches — all critical for preclinical and translational applications.
Take a deeper dive into the world of nanomedicine
Check our related content and discover how we can help
How we can help
TAMARA
Nanoparticle Formulation System
Fast & User-friendly operation
Rapid nanoparticle formulation with an intuitive workflow
Integrated R&D platform
Flexible working range (0.2-30 mL; 1 µg-5 mg of RNA per run)
Efficient use of materials
Reusable chips & zero formulation losses
Precision & Reproducibility
Microfluidic efficiency & precision