Polymer Nanoparticles (PNPs)
How are PNPs advancing nucleic acid therapeutics?
Polymeric nanoparticles (PNPs) expand the boundaries of nucleic acid delivery by offering unmatched chemical tunability and design flexibility. As RNA therapies grow more complex, PNPs are emerging as powerful alternatives — and complements — to lipid-based systems. Although polymer-based systems remain earlier in clinical development, they hold strong potential for expanded tissue targeting, improved stability, and prolonged or controlled release profiles.
What’s behind a PNP?
PNP composition & structure
Polymer nanoparticles are colloidal nanocarriers — typically ranging from ~10 to 1,000 nm — constructed from natural or synthetic polymers. Therapeutic cargos such as RNA, DNA, or small molecules can be encapsulated within the polymer matrix, confined in a core–shell structure, or adsorbed or conjugated to the particle surface, offering multiple strategies for payload protection and delivery.
Depending on their design and fabrication, polymeric nanoparticles can form nanospheres, where the cargo is uniformly distributed throughout the polymer matrix, or nanocapsules, in which the payload is enclosed within a polymeric shell. These systems are commonly built from polyesters—such as the synthetic polymers poly(lactic acid) (PLA) and poly(lactic-co-glycolic acid) (PLGA)—as well as amphiphilic block copolymers, including polyethylene glycol (PEG)–based copolymers that enhance stability and circulation time. Cationic polymers such as the synthetic polyamine polyethyleneimine (PEI) have also been extensively studied for nucleic acid condensation, although toxicity concerns have driven the development of safer alternatives. Natural polymers such as chitosan are also widely used for their biocompatibility and intrinsic bioactivity.
How do PNPs deliver RNA into cells?
Polymer–RNA interactions & Cellular uptake of PNPs
Many polymers used in PNP formulations are positively charged and can electrostatically bind negatively charged RNA, condensing it into compact nanoparticles that protect the payload from degradation. These polymers often contain combinations of primary, secondary, and tertiary amine groups, which support strong RNA binding while also enhancing interaction with the negatively charged cell membrane, facilitating cellular uptake through endocytic pathways.
Intracellular trafficking & endosomal escape
After internalization, PNPs are trafficked through endosomes, where the acidic environment triggers structural changes in the polymer. Protonatable amine groups can buffer the endosomal pH and destabilize the membrane, promoting release of RNA into the cytoplasm. This step is critical for all RNA modalities — whether mRNA, siRNA, or gene editing components — as therapeutic activity requires access to the cytosolic machinery. Consequently, polymer composition, charge density, and degradability are key determinants of successful intracellular RNA delivery.
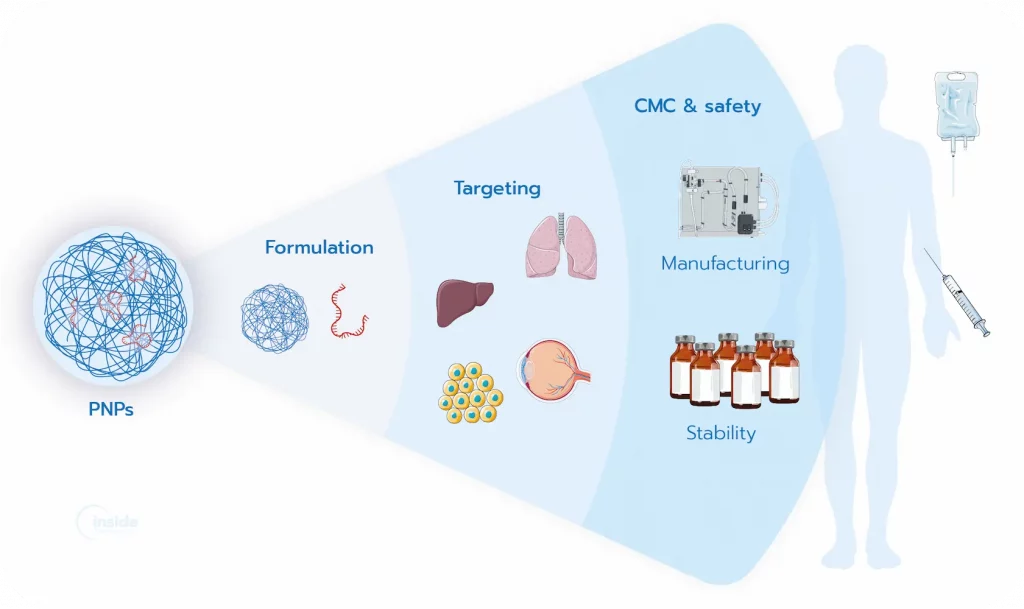
What are the strengths and challenges of PNPs?
PNPs offer unique advantages for delivering nucleic acids, making them a versatile alternative or complement to lipid-based systems.
Key advantages of PNPs
- Controlled & sustained RNA release – Tunable polymer composition and architecture allow tight control over particle size, degradation, and payload release, supporting chronic or sustained applications.
- Biocompatibility & stealth properties – Polymers can be engineered to reduce immune recognition, improve tolerability, and enable repeat dosing.
- Tailored delivery options – Versatile designs support specialized functions such as nuclear targeting, enhancing therapeutic reach.
- Stability & logistics – PNPs offer longer shelf life, robust storage, and simplified distribution for research and clinical applications.
- Application flexibility – PNPs are suitable for ex vivo and in vivo use.
Current limitations
- Complex formulation design – Developing optimized polymer structures for specific RNA cargos requires careful chemical design and testing.
- Limited field standardization – Testing, characterization, and reporting standards are still evolving.
- Scale-up – Manufacturing PNPs at global or clinical scale remains largely unproven.
- Preclinical development needs – Further research is required to fully assess safety, efficacy, reproducibility, and long-term performance.
Why do PNP characteristics matter?
Optimizing PNP formulations
The biological performance of polymeric nanoparticles depends heavily on careful formulation. Fine-tuning critical process parameters (CPPs) — including polymer type, molecular weight, composition ratios, solvent choice, and preparation conditions — is essential to achieve reproducible and effective PNPs. Optimized formulations enhance stability, maximize RNA delivery to target cells, and minimize toxicity, ensuring that each batch meets the desired performance standards.
Key PNP attributes
- Particle size and polydispersity (PDI): Influences cellular uptake and biodistribution.
- Surface charge: Affects interactions with serum proteins, cellular membranes, overall stability, and toxicity.
- Encapsulation efficiency: Impacts how much RNA is effectively delivered.
- Degradation & release profile: Controls the timing and duration of RNA release, supporting sustained or controlled therapeutic effects.
- Morphology: Shapes cellular uptake, intracellular trafficking, and payload release.
How are PNPs prepared?
PNP formulation methods
Conventional preparation methods include solvent evaporation, nanoprecipitation (or solvent displacement), emulsification/solvent diffusion, salting out, dialysis, and supercritical fluid technology. The choice of preparation method is crucial, as it allows fine-tuning of PNP properties to match the specific needs of each application.
Microfluidic synthesis of PNPs
To enhance control over PNP formation, microfluidic manufacturing has emerged as a powerful approach. In these systems, polymer and cargo solutions are combined under highly controlled flow, promoting rapid and reproducible self-assembly. Microfluidics allows better regulation of nanoprecipitation and the ability to screen multiple formulation conditions — such as flow rates, polymer composition, and mixing time — rapidly.
Unlike standard nanoprecipitation, where limited control over mixing can lead to variability in particle properties, microfluidics provides precise and tunable control. This precise mixing enables fine-tuning of particle size, narrow size distributions, and consistent encapsulation efficiency, all while minimizing material consumption.
Take a deeper dive into the world of nanomedicine
Check our related content and discover how we can help
How we can help
TAMARA
Nanoparticle Formulation System
Fast & User-friendly operation
Rapid nanoparticle formulation with an intuitive workflow
Integrated R&D platform
Flexible working range (0.2-30 mL)
Efficient use of materials
Reusable chips & zero formulation losses
Precision & Reproducibility
Microfluidic efficiency & precision