
Abstract
This guide outlines essential methods for lipid nanoparticles (LNPs) and liposomes characterization, detailing how factors like composition, size, surface charge, morphology, and encapsulation efficiency impact therapeutic delivery. It emphasizes quality control strategies to unlock the full potential of nanocarriers in precision medicine.
In the fast-paced biotechnology landscape, LNP characterization, and more generally characterization of lipid-based nanoparticles, such as liposomes and lipid nanoparticles (LNP) as become cornerstone for the efficient development of new therapies. LNP and Liposomes have emerged as versatile and promising tools for the delivery of genetic material and small molecules to target various locations within the body, consequently considering the pivotal role of their physicochemical parameters on the delivery and transcription efficiency, getting a fine understanding and control of the impact has become crucial.
Catalyzed by the remarkable achievement of Moderna’s Spikevax and Pfizer–BioNTech’s Comirnaty COVID-19 vaccines, both relying on the delivery of mRNA to the cell cytoplasm by an LNP, these novel delivery technologies have opened doors to a new generation of therapeutics.
However, the development of this new generation of precision medicine is accompanied by the challenge of increasing complexity, perfectly illustrated by the lipid nanoparticle. Specifically designed for efficient RNA (mRNA, siRNA, sgRNA…) encapsulation, their attributes – such as composition, size, surface charge, and encapsulation efficiency – significantly influence its therapeutics’ success, affecting both its stability all along the development process and the drug delivery and release efficiency.
This comprehensive review is dedicated to better understanding the distinctive roles and characterization methodologies for each of these LNP and liposome parameters, keeping in mind the goal of optimizing delivery efficiency to unlock their full potential.
Regulatory framework
To start with, it is important to mention that there is no clear regulatory framework that covers all the lipid-based nanoparticle types. In fact, this will mostly depend on the payload and application. For instance, while guidelines have been defined for regular vaccine therapies, by both the EMA and FDA, those current guidelines do not mention RNA therapeutics yet. Additionally, when it comes to biodistribution, requirements differ for RNA therapeutics and mRNA vaccines [1]. For these reasons, drug developers are highly advised to reach out to the relevant regulatory body before initiating preclinical studies.
Nevertheless, when it comes to organic nanoparticle’s characteristics, a set of good practices can be defined as a guideline, such as those derived from the “Liposomes Drug Products Guidance for Industry” guide provided by the FDA, [2] highlighting the importance of characterizing physicochemical Critical quality attributes (CQAs), including vesicle/particle size and size distribution, and morphology.
LNP and liposomes characterization: Chemical composition
Lipid-based nanoparticles are generally manufactured using self-assembly processes. Those processes, while very repeatable and controllable when using the appropriate synthesis method such as microfluidics, are not as well understood as historical top-down chemical synthesis processes. Therefore, any slight change in the synthesis process parameters can have major implications on the final nanoparticle characteristics. Let’s develop the most important ones here.
Typical Lipid Nanoparticles (LNP) composition
Nanoparticle synthesis generally involves the fast mixing of multiple ingredients, triggering the self-assembly of the particles.
In the case of LNP for instance, the most used lipid components are:
An ionizable/cationic lipid accounting for typically around 50 mol% of the LNP composition. Ionizable lipids are neutral at pH 7.0, they become positively charged at low pH, which ensures efficient nucleic acid encapsulation and delivery.
A helper lipid such as DSPC: a major component of the original lipid-based nanoparticle – such as liposomes – gave up its seat to the ionizable lipid in the case of LNP, and accounts for 10mol% of the current LNP composition.
A sterol lipid – generally cholesterol – whose role mostly depends on the associated compound, mostly helps with membrane stability and rigidity.
A PEG lipid at a small molar percentage (up to 3%), PEG is located on the surface of the LNP and has a major impact on numerous LNP characteristics such as size, stability, encapsulation efficiency…
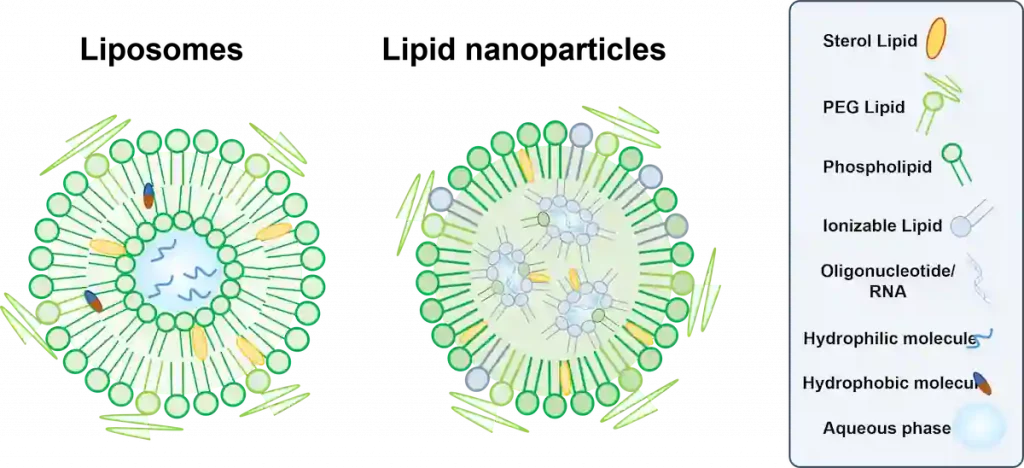
The ratio of each of the phases plays a major importance to ensure the stability and efficiency of the final drug product. Indeed, small changes in mixing ratio can lead to significantly different compositions and physicochemical parameters of the nanoparticle, thus leading to very different delivery efficiencies within the cells.
For instance, the lack of Pegylated lipid will lead to an increase in recognition by the human immune system – due to a decrease in stealth effect – while greatly increasing the final nanoparticle size, highly affecting its delivery capabilities.
Composition characterization tools
Nanoparticle composition is generally assumed by design from the ratio of compound used in the self-assembly process. Nevertheless, they can also be characterized using typical analytical chemistry tools such as HPLC or UHPLC and are generally expressed in mg/mL and ratios of the phases.
LNP and liposomes characterization: Physicochemical properties
The physicochemical properties of lipid-based nanoparticles hold major significance as they influence their capacity to encapsulate, retain, and properly deliver therapeutic substances.
The subsequent set of characteristics typically serves as valuable parameters for characterizing lipid-based nanoparticle formulation. Fluctuations in these attributes could potentially result in alterations to the quality of the therapeutics, potentially causing lowered delivery, transcription efficiency, or increased drug leakage from the liposomes or LNP.
LNP and liposomes size characterization
Particle size is one of the most critical nanoparticle parameters to be characterized due to its impact on drug delivery efficiency and should thus be tailored depending on the delivery route and the target cell.
In practice, particles that are too small are either cleared from the bloodstream by the kidney before being delivered or can induce toxicity to the target cells. On the other hand, too large nanoparticles struggle to penetrate cells, which greatly reduces cell uptake. [3]
Typically, within 50 to 200 nm, sought nanoparticle size will depend on its type and target cell.
For instance, the LNP target size will be 80-100 nm for optimal delivery within cells, while the optimal nanoparticle size will be smaller – usually in the order of 60 nm – for delivery to cancer cells. Learn more about it in our passive nanoparticle targeting review.
Parameters influencing LNP and liposome size
The first parameter impacting the final lipid-based nanoparticle size is its composition. As previously mentioned, differences in the ratio of the different lipids will lead to different composition and thus size. In practice, PEG seems to have the most impact on size: M. Lokugamage et al. [4] showed that PEG-less lipid-based nanoparticles were not only much bigger (>200nm) than those with as low as 0.5%mol PEG (around 80nm) but also polydisperse and less stable.
It is also important to note, somewhat surprisingly, that the size of lipid nanoparticles is not affected by the size of the RNA cargo, as described in the study: A careful look at lipid nanoparticle characterization: analysis of benchmark formulations for encapsulation of RNA cargo size gradient
Additionally, as various synthesis methods are available, nanoparticle final size will greatly depend on the nanoparticle manufacturing methods and synthesis parameters used. Considering the importance of nanoparticle size on drug delivery efficiency, drug developers should ensure that they use a manufacturing process that allows them good control of this parameter all along the development process.
Additionally, batch-to-batch repeatability is critical as unexpected changes in size could signal issues with manufacturing processes.
For those reasons, microfluidics is currently considered the gold standard by nanoparticle synthesis specialists thanks to its unique ability to reproduce synthesis conditions, thus final size, while being extremely repeatable.
Nanoparticle size characterization tools
Numerous methods are currently available for the characterization of LNP and liposomes size. However, due to the difficulty of obtaining reliable measurements at that stage, nanoparticle specialists usually rely on a combination of different techniques to ensure optimal measurements of the particle size.
The most used tools for liposomes and LNP size characterization are:
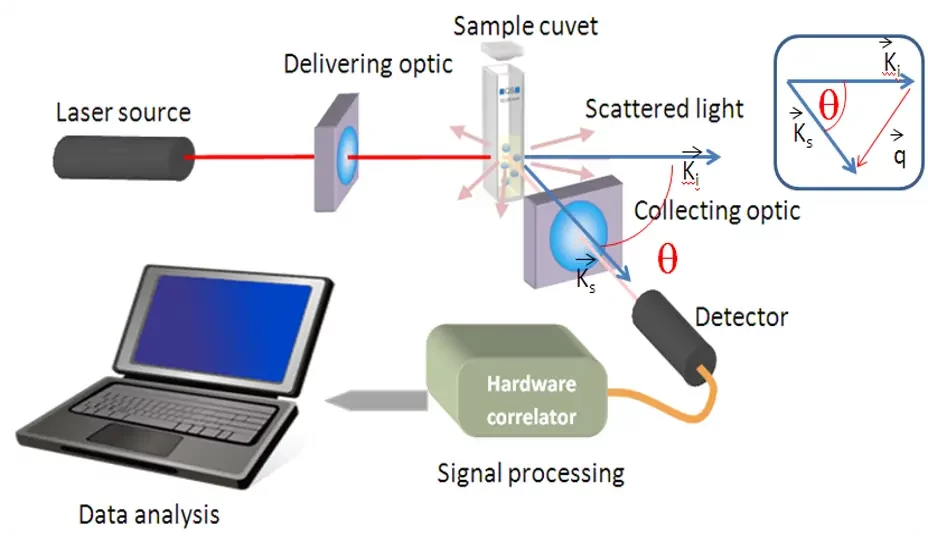
- DLS (Dynamic light scattering) relies on light scattering through the nanoparticle suspension to measure mainly size and polydispersity index as described in the below schematic courtesy of Cordouan Technology’s.
Overall, DLS is excellent for checking qualitative parameters (i.e batch to batch reproducibility) but sometimes lack of the require absolute accuracy. To learn more on DLS check Cordouans’ website. - MADLS (Multiangled dynamic light scattering): Advanced version of the DLS tool, it offers better resolution and the ability to identify subpopulation, however at a higher cost.
- TRPS (Tunable Resistive Pulse Sensing) retrieves the particle size by measuring in real-time the impedance of the nanoparticles going through a set of nanopores. In addition to this, TRPS also provided concentration measurements.
- NTA (Nanoparticle Tracking Analysis): Based on Particle Tracking Velocimetry, it relates the rate of Brownian motion to nanoparticle size. While NTA is a good tool for characterizing nanoparticle size, the method is slower and less reproducible than DLS
- Cryo-TEM (Transmission Electron Microscopy): Ultimate tool for nanoparticle size measurement as it allows for direct observation of their size and structure. However, it is very time-consuming, requires high user expertise and shows only observations on part of the sample.
Lipid-based nanoparticle size dispersity: Polydispersity Index
Closely related to the size, polydispersity index or PDI, characterizes the width or spread of the nanoparticle size distribution profile.
PDI is defined by the square of the standard deviation of the nanoparticle population over the nanoparticle mean diameter.
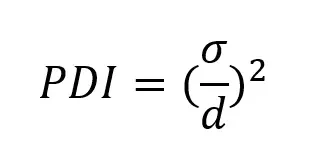
PDI should therefore be as close to 0 as possible for the most uniform population.
FDA’s guidelines suggest that PDI should remain below 0.3. For this reason, typical target PDI values should range from 0.05 to 0.2 to keep some flexibility and error margin. In practice, large PDI indicated a too heterogeneous nanoparticle population that will show poor transcription efficiency and repeatability.
Parameters influencing LNP and liposome’s PDI
As for the size, both composition and manufacturing processes have a major impact on the final nanoparticle polydispersity.
First, the nature of the synthesized nanoparticle will affect the PDI: some types of nanoparticles will naturally be more stable than others, offering lower PDI.
When it comes to manufacturing, historical processes such as thin film hydration or high-pressure homogenization tend to show very high PDI in the order of 0.5 or more, depending on the composition.
For this reason, microfluidics-based methods should be preferred as they offer the highest nanoparticle population uniformity thanks to their unique ability to perfectly reproduce nanoparticle synthesis conditions all along the manufacturing process.
Nanoparticle PDI characterization tools
Likewise, characterization tools for size and PDI are very similar. In the case of PDI, batch methods such as DLS or TRPS should be preferred to characterize the whole nanoparticle population at once.
LNP Zeta potential
The zeta potential corresponds to the measure of the effective electric charge close to the nanoparticle’s surface. Often mistaken as the surface charge of the particle, it remains an important parameter to measure as it is a good indicator of both the stability of the nanoparticle suspension and its potential side effects.
In practice, a high value of zeta potential indicates a good stability of the nanoparticle suspension (larger than ± 30 mV is considered moderate stability), while an almost neutral zeta potential will lead to agglomeration and potential coalescence. On the other hand, positive charges too high close to the surface of the LNP will lead to an increased toxicity. Therefore, for an optimal stability with a minimized toxicity, manufactured drugs should seek a zeta potential in the order of ±30 mV or use stabilizing agents such as PEG to improve stability even at low ZP values.
Parameters influencing LNP and liposome’s Zeta potential
Composition and pH are the most important parameters that influence zeta potential. For example, a decrease in pH of the solution will increase the positive charges on the particle surface leading to a zeta potential will increase.
Zeta potential characterization tools
Most of the methods used for size and PDI characterization of liposomes and LNP have also been adapted for Zeta potential measurements:
- Electrophoretic Light scattering uses the same measurement principle as DLS measurement systems. However, instead of measuring Brownian motion it uses an oscillating electric field to cause particle motion, which is dependent on particle charges.
- TRPS: based on impedance measurement, TRPS offers the ability to measure individual particle Zeta potential, which is proportional to the pulse duration, while carrying out size measurement, making it very time efficient.
Lipid based nanoparticles Morphology
In addition to the size related parameters, and surface charge, it is also important to understand more precisely the surface and internal organization of the nanoparticles to get a better understanding of their drug delivery capabilities.
For instance, even the simplest nanoparticle structure, the liposome, can have its encapsulation properties totally changed depending on its internal organization.
Even though liposomes are relatively simple in composition – they are generally made of majority of a single amphipathic lipid type with some helpers – their internal organization can vary from the most basic ones – a single unilamellar bilayer – to very complex multilamellar or “onion-like” structures.
These various types of structure also differ in their encapsulation capabilities: Multilayered liposomes can for instance encapsulate multiple cargoes, hydrophilic or lipophilic, of different size within one single structure, allowing for the simultaneous delivery of multiple compounds to the same location.

Parameters influencing LNP and liposome’s morphology
As for the previously discussed parameters, lipid-based nanoparticle internal organization will vary depending on their type: Therefore, both their composition, cargos and synthesis method will play a role in their internal organization.
Nanoparticle morphology characterization tools
Unlike for size or encapsulation efficiency measurements, bulk-based technics cannot be used for characterization of the LNP and liposomes morphologies, as those techniques only provide averaged information about the sample properties.
To do so, single-particle analytical methods, such as Transmission Electron Microscopy (TEM), Scanning Electron Microscopy (SEM), X-Ray diffraction or Atomic Force Microscopy (AFM) should be used.
However, LNP, liposomes as well as all other organic nanoparticles are “soft” materials usually in liquid suspensions, which makes them difficult to characterize with some of the microscopy techniques. TEM and SEM are particularly suitable for inorganic and solid nanoparticles, as they can withstand the measurement conditions (under vacuum, highly focused electron beam…), unlike LNP and liposomes which require extra care in the drying process as well as the use of contrast agents.
Most used characterization method for liposomes and LNP characterization is Cryo-TEM. In this technique, nanoparticles are frozen in liquid nitrogen, to prevent the formation of ice crystals, and are put directly under direct TEM observation.
This allows an in-depth understanding of the surface morphology and inner structure of the nanoparticles, as well as better understanding of the actual particle size population.
Liposomes and LNP Encapsulation Efficiency
Unlike the previously discussed parameters, the encapsulation efficiency, or EE%, does not relate only to the lipid-based nanoparticle intrinsic characteristics, but to its ability to encapsulate cargo within it, aka, its efficiency.
Encapsulation efficiency is generally calculated as the ratio of encapsulated cargo over the initially injected cargo.
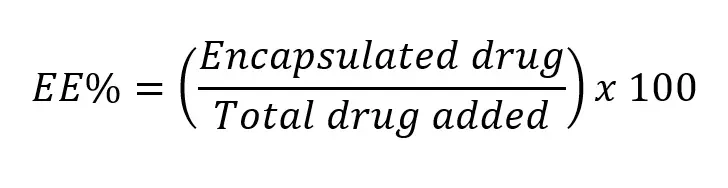
Target encapsulation efficiency will obviously vary depending on the nanoparticle types and cargo. Its values can reach 95% or more for mRNA encapsulation into the latest generation of LNP but can also be much lower for other compounds such as gold nanoparticles into liposomes for instance.
Parameters influencing Lipid based nanoparticle encapsulation efficiency
Encapsulation efficiency is one of the parameters where the synthesis process has the most importance: For the exact same nanoparticle type and cargo, the nanoparticle encapsulation efficiency can nearly vary from one order of magnitude depending on the used manufacturing process.
Currently, the highest encapsulation efficiencies are achieved with microfluidics-based synthesis process, where the EE% can reach up to 95% for RNA into LNP.

Accelerate your formulation with our
LNP Formulation system
Encapsulation Efficiency characterization tools
Numerous methods are available for the measurement of the liposomes and LNP encapsulation efficiency, and their choice will greatly depend on the encapsulated cargo. As the total amount of drug usually corresponds to the initial amount of drug added during the fabrication process, the main unknown is the amount of encapsulated drug.
Several encapsulation efficiency measurement methods are available, let’s explore the most used ones:
- Dialysis : In this method, the free drug is separated from carrier and measured. The encapsulated drug is indirectly calculated as : total amount of drug – free drug.The characterization can be done after the dialysis phase. The free drugs being separated from the encapsulated drug by the semi permeable membrane, it can be measured – through concentration – by what is remaining in the dialysis buffer. However, this method is relatively time consuming – 2 to 24 hours – and dialysis is known to impact the physico-chemical characteristics of the LNP. Other methods of separation such as centrifugation or tangential flow filtration (TFF) can be used to isolate the free drugs.
- Fluorescence: For RNA-LNP EE% determinization, fluorescence is generally used Typically, once the free RNA has been isolated, a fluorescent tag – such as the so-called “RiboGreen” reagent – is conjugated to each RNA and the fluorescence intensity, measured by a fluorometer, scales with the concentration of RNA. Alternatively, a UV-Vis spectrometer can be used to measure the characteristic absorbance peak of the nucleic acids at 260 nm. However, the latter method has lower resolution and specificity than the fluorescence assay.
Main takeaway on Liposomes and LNP characterization
To conclude, the emergence of lipid-based nanoparticles, exemplified by liposomes and lipid nanoparticles (LNPs), has ignited a new era in the delivery of biological material. However, as we venture into this new frontier, we are met with the intricate challenges posed by the growing complexity of these novel delivery technologies.
While not yet fully framed by regulation, this increased complexity led to the definition of a new set of critical nanoparticle characteristics to be evaluated including size, size dispersity surface charge, morphology, and encapsulation efficiency.
Throughout this comprehensive review, our focus has been directed towards understanding the role of each parameter, the parameters affecting them most and the methods to characterize them.
From our investigation, it becomes evident that nearly all these parameters stem directly from the composition and manufacturing process of the nanoparticles. As a result, prudent selection of nanoparticle composition and manufacturing processes is imperative to fully harness the potential of nanoparticle-mediated delivery.
In conclusion, we have determined that a more profound characterization and comprehension of these parameters, along with their interplay with biological factors, is essential for unlocking the complete capabilities of liposomes and LNP.

Looking to get started with RNA-LNP formulation?
Reach out to us to learn how we can help!
References
[1] Vervaeke, P et al. “Regulatory guidelines and preclinical tools to study the biodistribution of RNA therapeutics.” Advanced drug delivery reviews vol. 184 (2022): 114236. doi:10.1016/j.addr.2022.114236
[2] Liposome Drug Products: Chemistry, Manufacturing, and Controls; Human Pharmacokinetics and Bioavailability; and Labeling Documentation: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/liposome-drug-products-chemistry-manufacturing-and-controls-human-pharmacokinetics-and
[3] Danaei, M et al. “Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems.” Pharmaceutics vol. 10,2 57. 18 May. 2018, doi:10.3390/pharmaceutics10020057
[4] Lokugamage, Melissa P et al. “Optimization of lipid nanoparticles for the delivery of nebulized therapeutic mRNA to the lungs.” Nature biomedical engineering vol. 5,9 (2021): 1059-1068. doi:10.1038/s41551-021-00786-x








