
mRNA LNP Revolution: Unlocking New Frontiers in Pharmaceuticals, Vaccines, and Therapeutics
Abstract
mRNA–LNP (Lipid nanoparticles) are redefining modern therapeutics by enabling precise, safe, and efficient delivery of genetic instructions. First validated by COVID-19 vaccines, this versatile platform now drives advances in treatments for rare diseases, cancer, cardiovascular and neurodegenerative disorders, and CRISPR-based gene editing. This review introduces RNA–LNP technology, detailing its composition, production workflow, and emerging applications.
mRNA LNP is a promising approach to a wide range of novel pharmaceuticals, vaccines, and therapeutic solutions. Its groundbreaking potential became evident to the public during the COVID-19 pandemic with the astonishingly quick development of the mRNA LNP Vaccine.
This innovative RNA technology combining Messenger ribonucleic acid (mRNA) and lipid nanoparticle (LNP) holds the unique potential to transform the biotech and pharmaceutical landscape by unlocking numerous new possibilities of treatments, not only in the infectious disease field but also for rare diseases or cancer treatment…
In this article, we will explore what makes mRNA LNPs so unique by delving into their nature, working principle, manufacturing process, and the numerous applications that are called to change the future of medicine.
What are mRNA LNP ?
mRNA-LNPs are spherical particles of size ranging from 50 to 200 nm, generally made up of five main components: The cargo (mRNA) and the 4 lipids component of the LNP:
- mRNA: messenger RNA is the cargo of the mRNA LNP complex. This single-strand RNA molecule corresponds to the nucleotide sequence of a gene, which is used for the synthesis of a protein through transcription by a ribosome.
- Phospholipids: Phospholipids are amphipathic, meaning that they have both hydrophilic (water-loving) and hydrophobic (water-hating) regions. Due to this characteristic, those lipids are mostly located in the nanoparticle membrane and help improve encapsulation efficiency.
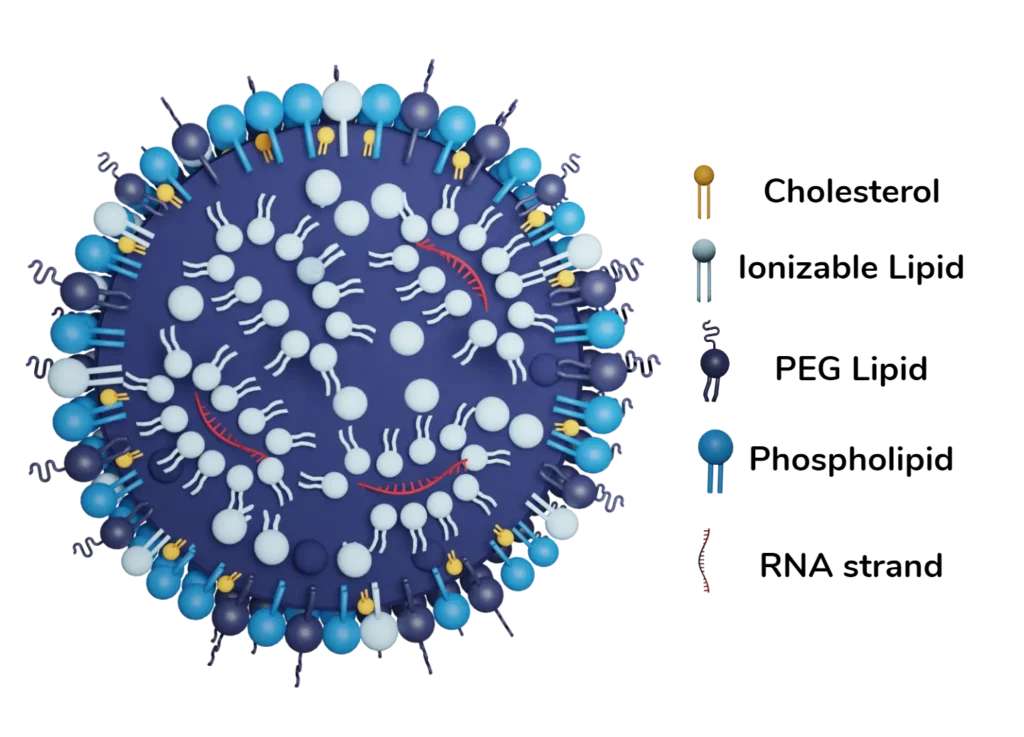
- Sterol lipids: Cholesterol is added to the lipid bilayer to increase its stability, and rigidity and help decrease drug leakage. Cholesterol also helps to protect the cargo from degradation
- PEGylated lipids: although a minor fraction of the lipid composition, have a significant impact on nanoparticle size, circulation duration, and stability.
Primarily situated on the nanoparticle’s surface, their long tail helps contribute to the nanoparticle’s structural integrity. - Ionizable cationic lipids: They are the core component of the complex as they constitute about 50% of it and are critical for encapsulating the RNA cargo.
Lipid nanoparticles have this unique capability of being neutral at pH 7 but become positively charged at lower pH. They serve three vital functions in the nanoparticle structure:- 1/ They improve oligonucleotide encapsulation during LNP fabrication
- 2/ They enhance the cell uptake of the nucleic acid delivery
- 3/ They help reduce toxicity at neutral pH.
Due to their cornerstone role in facilitating mRNA delivery through lipid nanoparticles (LNPs), ionizable lipid has emerged as a central focal point in the ongoing patent disputes between pharmaceutical and biotechnology companies engaged in the development of novel mRNA medicines, each aiming at developing their patented ionizable lipid, to get access to the proprietary LNP.
Learn more in our review on ionizable lipids for LNP.
mRNA LNP working principle
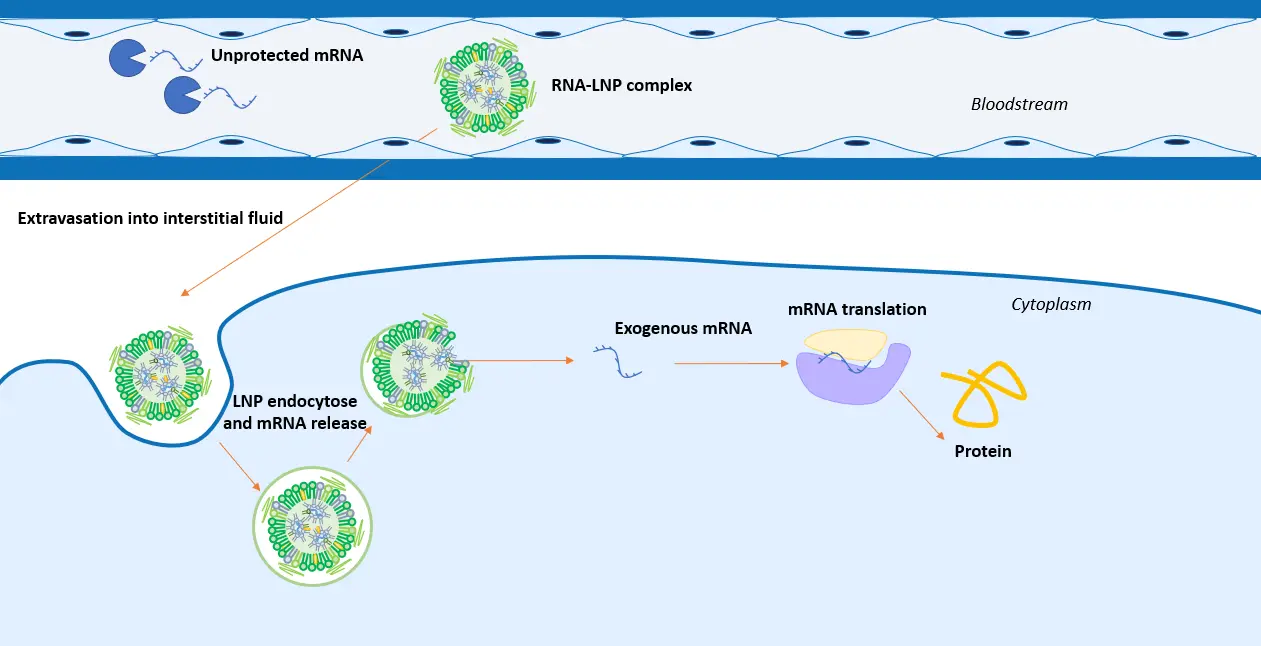
Upon introduction into the body, mRNA, and more generally all nucleic acids, are susceptible to degradation by serum endonucleases. To protect them, nucleic acids therapeutics have traditionally relied on viral delivery methods, such as inactivated viruses, for nucleic acid delivery, but they exhibited limitations like side effects and poor loading capacity.
In parallel to this, over the past 50 years, lipid-based nanoparticles, such as liposomes, have been used for drug and small-particle delivery. However, their simple structure hindered efficient nucleic acid delivery and encapsulation while causing high toxicity.
For this reason, a specific type of nanoparticle specialized in oligonucleotide transports has been engineered: The LNP.
Their unique structure, including an ionizable lipid, allows for a high encapsulation rate while ensuring efficient delivery of the nucleic acids.
The intracellular mechanism of mRNA delivery by lipid nanoparticles relies on several factors:
First, encapsulating mRNA into LNP improves circulation time by preventing its degradation. Additionnaly, it binds to a cell, the LNP will help the nucleic acid cross the cell barrier via the endocytosis process. Finally, the drop of pH happening during the endocytosis will lead to a degradation of the RNA LNP structure, liberating the cargo into the cytoplasm.
Manufacturing process
The process of preparing mRNA-LNP is intricate and involves several distinct steps:
mRNA identification
Initially, the process begins with the identification of the right mRNA sequence that encodes the targeted protein. This sequence is encapsulated by its head and tail, mimicking an endogenous mRNA structure. This simulation aims to optimize gene expression and enhance stability.
Once the right mRNA sequence is identified, it must be produced and purified before being encapsulated into the LNP.
mRNA Production and Purification
RNA can be generated through three primary methods:
- Chemical Synthesis: This method is the simplest, most continuous, and cost-effective. However, it is suitable only for short RNAs (<100 nucleotides) and is not ideal for long oligonucleotides.
- Host Cell (Recombinant Production): Despite recent scientific advancements in preventing RNA degradation, using a host cell (e.g., E. coli) for recombinant production lacks scalable manufacturing solutions.
- Enzymatic Synthesis: Two approaches in enzymatic synthesis are gaining prominence: Polymerase Chain Reaction (PCR) coupled with Polymerase Chain Transcription (PCT) and advanced in vitro transcription (IVT). While PCT requires further improvement, IVT has become the gold standard for mRNA manufacturing. A DNA template – usually a plasmid DNA – codes for the protein of interest, transcribed into an RNA strand by an enzyme, and then purified.
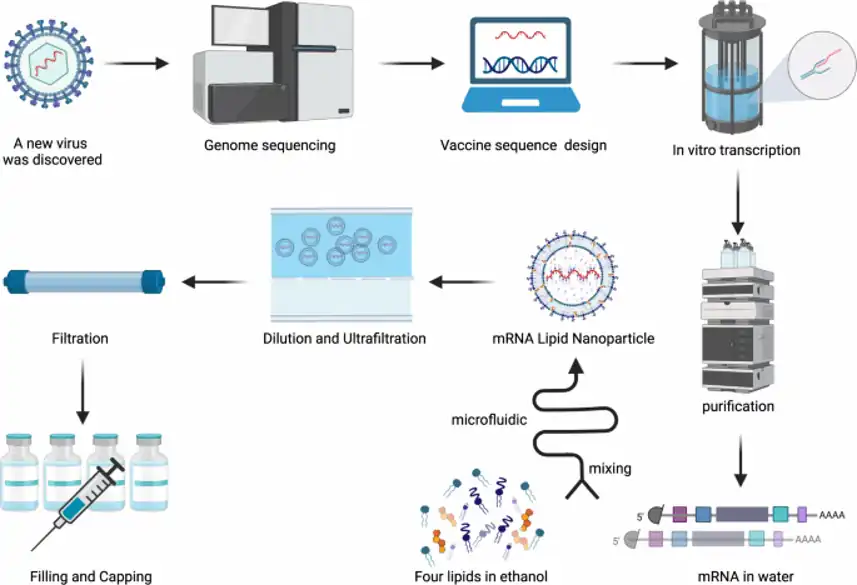
Encapsulation into Nanoparticles
As described above, once manufactured, the mRNA strand should be encapsulated into an LNP for efficient intracellular delivery.
mRNA LNP manufacturing process
Traditionally, lipid-based nanoparticles were produced in batches through high-energy methods such as thin-film hydration, high-pressure homogenization, and sonication. However, these LNP manufacturing methods exhibited poor repeatability, encapsulation efficiency, and yield, rendering them unsuitable for mRNA vaccine development.
In response, mRNA-LNP is now manufactured using self-assembly methods. Lipids are dissolved in a solvent, typically ethanol, while a low-pH water solution containing mRNA is an antisolvent. During mixing, the solutions blend, reducing lipid solubility and triggering the self-assembly of nanoparticles.
Given the significant impact of mixing conditions on nanoparticle characteristics (size, charge, etc.), microfluidics has emerged as the most utilized method for mRNA-LNP manufacturing. Its unique ability to replicate mixing conditions, coupled with scalability from microliters to liters, makes it the preferred choice in the production process.
To complete the process, mRNA LNP purification is then carried out using standard methods such as dialysis or tangential flow focusing (TFF).
LNP Formulation
As introduced in our review on the optimization of mRNA LNP formulation, in addition to the manufacturing process, numerous parameters of the lipid nanoparticle play a role in the transfection efficiency, including lipid composition, lipid ratios, size, pH, dialysis process…
mRNA LNP applications
mRNA therapeutics have a wide range of potential applications, including:
- mRNA Vaccines for infectious diseases: mRNA-LNPs have been used to develop highly effective prophylactic vaccines against a variety of infectious diseases, including COVID-19, influenza, and malaria. In the context of vaccines, they are used to deliver mRNA that encodes the antigens of the target pathogen, such as the spike protein of the COVID-19 virus for the covid 19 vaccine. Once the mRNA is delivered to cells, the release of this protein triggers an immune response against the pathogen and helps to protect the patient from infection. As of July 2021, there was a total of 73. prophylactic vaccines currently in preclinical or clinical trials, and 3 already approved. (1)
- Cancer immunotherapy/vaccines: When it comes to cancer, mRNA LNP are generally used as therapeutics vaccines. They work by delivering an mRNA that encodes chimeric antigen receptor (CAR) T cells. CAR T cells are engineered T cells that express a chimeric antigen receptor on their surface. This receptor allows the CAR T cells to recognize and kill cancer cells. mRNA-LNPs have been used to deliver CAR T cell mRNA to patients with leukemia and lymphoma, with promising results.
- Gene therapy: Additionally, LNP delivery of mRNA can be used to introduce an RNA strand that encodes therapeutic proteins to treat genetic diseases. For example, mRNA-LNPs are being investigated for the treatment of Duchenne muscular dystrophy, cystic fibrosis, and sickle cell disease. In gene therapy, the goal is to deliver mRNA that encodes a functional copy of the gene that is mutated in the disease. Once the mRNA is delivered to cells, it is translated into a protein that can compensate for the defective protein.
- CRISPR-Cas9 gene editing: In the context of genome editing, an mRNA strand that encodes for a CRISPR-Cas9 gene-editing machinery is delivered to the cell. CRISPR-Cas9 is a powerful tool that can be used to make precise changes to DNA. mRNA-LNPs have been used to deliver CRISPR-Cas9 machinery to cells to correct genetic mutations that cause a variety of diseases, including sickle cell disease and beta-thalassemia.
Conclusion
mRNA-LNPs are a promising new approach to drug delivery. They have several advantages over other drug delivery systems, including high efficiency, low toxicity, and versatility. mRNA-LNPs have the potential to be used to develop a wide range of new and effective therapies for a variety of diseases.
In addition to the applications mentioned above, mRNA-LNPs are also being investigated for use in other areas, such as Protein replacement therapy Treatment of autoimmune diseases Treatment of neurodegenerative diseases Treatment of cardiovascular diseases. More generally, the RNA-LNP technology can also be used for the delivery of other types of nucleic acids such as siRNA, miRNA… RNA therapeutics are still in the early stages of development, but it has the potential to revolutionize the way that many diseases are treated.

Looking to get started with RNA-LNP formulation?
Reach out to us to learn how we can help!







