An introduction to LNP or Lipid nanoparticle for siRNA delivery
Over the past few years, the RNA delivery field has undergone significant changes with the advent of Lipid nanoparticles or LNP. In this context, the use of lipid nanoparticles for siRNA delivery has become a key game-changer for the whole gene silencing and gene therapy field, opening doors to new treatments for rare diseases.
To be more precise Lipid nanoparticles, or LNP, are submicron particles specifically engineered to protect nucleic acid from degradation and optimal RNA delivery to the cells. Beyond their well-documented role in mRNA-based vaccines, LNPs possess versatility in transporting various oligonucleotides, such as siRNA, also known as small interfering RNA or silencing RNA.
Indeed, siRNA is a pivotal component of the RNA interference process (RNAi), thanks to its ability to selectively silence genes, offering potential solutions to previously untreatable conditions. This makes siRNA the leading candidate for the novel generation of genomic medicines.[3] In this context, LNP is being used as an alternative solution to AAV or other types of non-viral delivery systems, for the delivery of siRNA, as it permits unlocking several of the main bottlenecks of the current gene therapies including: large-scale manufacturing, lowered toxicity and improved batch-to-batch reproducibility.
We will start with an illustration of its role in gene regulation, as well as its potential applications. Next, we will focus on the crucial aspect of its delivery, discussing the limitations faced by traditional delivery methods and highlighting the emergence of lipid nanoparticles as a promising solution. Finally, we will get an insight into the siRNA-LNP manufacturing process
SiRNA working mechanism
siRNA is a type of non-coding double-stranded RNA molecule, first discovered in plants in 1999 [1], then in mammalian cells. Typically around 21-24 bases pairs in length, it participates to the RNA interference (RNAi) process by specifically targeting and silencing genes, making it an invaluable tool for studying gene function and developing potential RNA therapeutic interventions.
siRNA mechanism involves four steps that ultimately prevent mRNA from being translated into proteins [2]:*
1/ Formation or delivery
The process begins with the introduction of an exogenous siRNA molecule to the cell cytoplasm (e.g synthetic delivered via a lipid nanoparticle) or the generation of endogenous double stranded silencing RNA through the slicing of the dsRNA (double-strand RNA) by the Dicer enzyme.
2/ Loading into the RNA-Induced Silencing Complex (RISC)
These siRNA molecules are incorporated into the RNA-induced silencing complex (RISC), which contains Argonaute proteins, before being unwounded, to only keep one strand called the guide strand. The RISC complex helps stabilize the guide strand and positions it for interaction with target mRNA molecule.
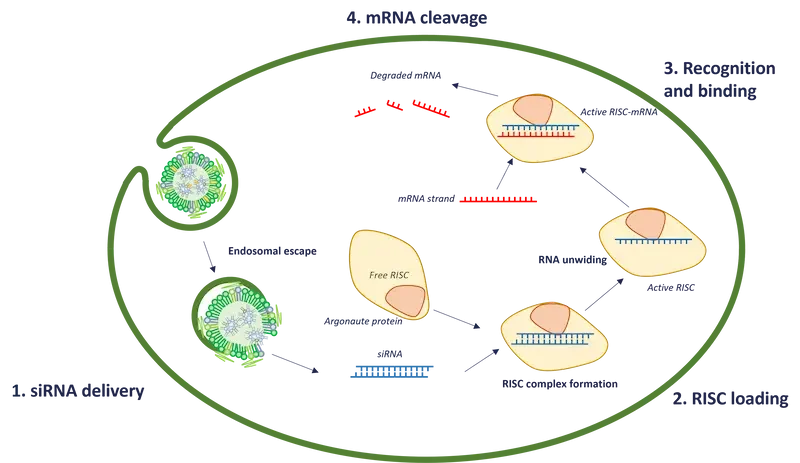
3/ Recognition and Binding
The guide strand of the silencing RNA contains a sequence complementary to the target mRNA's sequence. This enables it to recognize and bind to the mRNA through base-pairing interactions. The binding occurs primarily in the 3' untranslated region (UTR) of the mRNA.
4/ mRNA Cleavage or Translational Repression
Depending on the degree of complementarity between the silencing RNA and the target mRNA, two main mechanisms can occur:
- Perfect Complementarity: If the they both exhibit perfect complementarity, the RISC complex catalyzes the cleavage of the target mRNA within the binding site. This leads to rapid degradation of the mRNA, preventing protein synthesis.
- Partial Complementarity: In cases of partial complementarity, the RISC complex does not induce mRNA cleavage but instead initiates translational repression. This inhibits the translation of the target mRNA into protein by preventing ribosome binding or elongation.
This powerful ability to selectively inhibit or suppress specific gene expression has opened up new broad fields of research with the potential to revolutionize medicine.
Silencing RNA Delivery process
As we understood its great potential for gene exploiting, numerous challenges to fully exploit it remain. One of the most important ones is achieving efficient delivery into target cells or tissues.
The negatively charged nature of these small oligonucleotides makes it difficult for them to cross cell membranes on their own. Several approaches have been developed to overcome these delivery challenges, including viral vectors, lipid-based nanoparticles, and chemical modifications.
Viral vectors are one of the most used delivery systems in the drug industry. These vectors can efficiently deliver siRNA into cells by exploiting their natural ability to infect host cells. However, concerns regarding safety and immunogenicity have limited their widespread use in clinical applications. As a result, researchers have turned their attention towards non-viral delivery systems such as lipid-based nanoparticles.
Why use lipid nanoparticles for siRNA delivery ?
Lipid nanoparticles (LNP) offer several advantages over viral vectors, including ease of production, low immunogenicity [2], and tunable properties for efficient siRNA delivery. These nanoparticles can encapsulate these oligonucleotides and protect them from degradation in the extracellular environment, ensuring effective delivery of silencing RNA into the cytoplasm where it exerts its gene-silencing effects. Chemical modifications of silencing RNA molecules themselves also play a crucial role in improving stability and enhancing delivery efficiency.
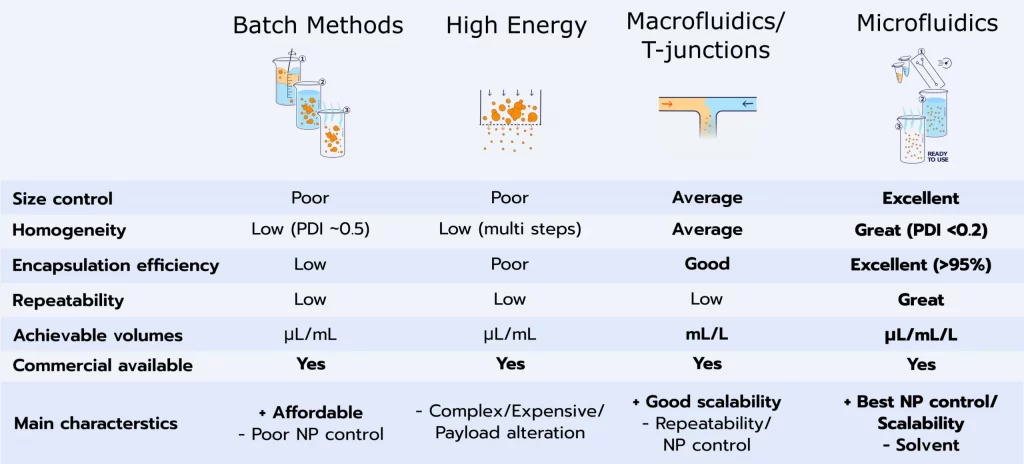
Numerous methods are available for the synthesis of these siRNA-LNP complex, including high-pressure homogenization, thin film hydration, and sonication... however, all those methods suffer from poor critical attributes control, reproducibility, and show major difficulties to scale-up, making them inappropriate for drug delivery.
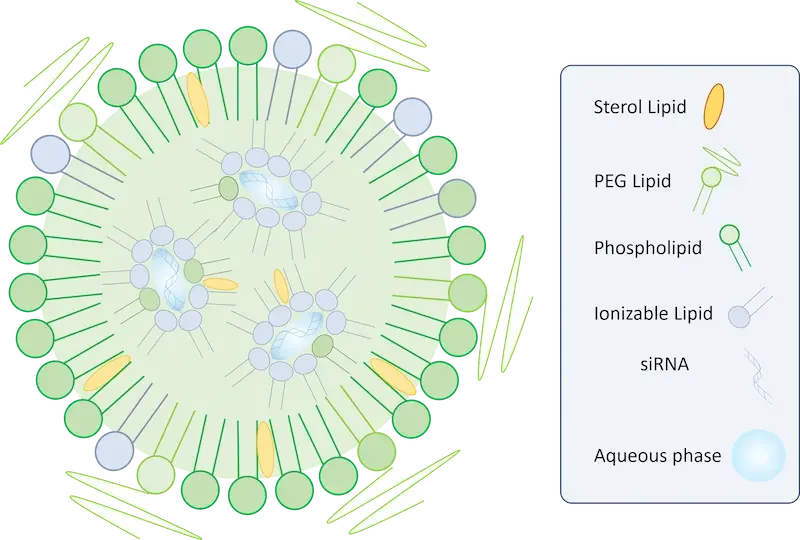
Due to these drawbacks, those RNA-LNPs are currently produced using self-assembly techniques involving the mixing of two solutions: a solvent, typically ethanol in which lipids are dissolved, and an antisolvent, commonly water-containing the nucleic acid. When these solutions are mixed, they form a blended mixture, causing a decrease in the solubility of lipids in their solvent. Consequently, this triggers the spontaneous self-assembly of nanoparticles.
Considering the importance of the mixing process on the ultimate characteristics of the nanoparticles, such as size and charge, cannot be overlooked. To address this concern, microfluidics has emerged as the preferred method for manufacturing RNA-loaded LNP. The attractiveness of microfluidics lies in its distinct ability to replicate and control the precise microfluidic mixing conditions critical for RNA-LNP synthesis and its capability to scale up the production from microliters to liters, making it the most widely utilized approach for producing those RNA-LNP complexes.
Typical application
After understanding the basics of the RNA interference process via silencing RNA, let's get a sneak peak to its various applications
Molecular Biology
In molecular biology, a prominent application lies in gene silencing. This involves targeting specific genes, thereby reducing or completely silencing their expression levels. Such precision allows researchers to investigate the impacts of gene expression and functions, providing valuable insights into disease pathways.
Gene therapies
The unique gene silencing capabilities of siRNA have paved the way for applications in gene therapies. By crafting oligonucleotide sequences complementary to the mRNA of disease-associated genes, it becomes possible to temporarily and selectively inhibit their expression. This targeted intervention holds immense promise for slowing down or halting disease progression, offering a groundbreaking approach to the development of RNA therapeutics. The potential applications span a wide range of diseases, including cancer, viral infections, and genetic disorders. This method has demonstrated its clinical and commercial viability, evidenced by Alnylam's successful release of the ONPATTRO siRNA-LNP vaccine in 2018.
Agricultural research
Beyond medical applications, RNA interference technologies have found utility in agricultural research for crop improvement. In this context, siRNA molecules are designed to target specific genes related to plant growth or defense mechanisms. By manipulating gene expression, researchers can enhance crop yield or bolster resistance against pests or diseases. This innovative use of RNA interference contributes to advancing sustainable agricultural practices and addressing challenges in food security.
Upcoming challenges for siRNA gene therapies
Aside from using lipid nanoparticles for siRNA delivery to improve its efficiency, other challenges should be overcome to fully exploit the potential of this technic.
The first remaining technical challenge of silencing RNA research is off-target effects. While those oligonucleotides are designed to specifically target a particular gene, there is always a possibility that they may also silence unintended genes with similar sequences. This can lead to unpredictable outcomes and hinder the accurate interpretation of experimental results.
In addition to off-target effects and delivery challenges, another obstacle is the potential for immune responses. When an siRNA drug is introduced into the body, foreign nucleic acids can trigger an immune response, leading to inflammation or even cell death. This immune response not only affects the efficacy of gene therapy but also poses safety concerns for clinical applications. Developing strategies to minimize immune responses while maintaining therapeutic effectiveness is crucial for advancing novel gene therapies.
Looking to synthesize Lipid nanoparticles for siRNA delivery? Shoot us a message!
References
-
Hamilton, A., et al. A Species of Small Antisense RNA in Posttranscriptional Gene Silencing in Plants (1999). DOI: 10.1126/science.286.5441.950
-
Kalita, T., et al. siRNA Functionalized Lipid Nanoparticles (LNPs) in Management of Diseases
DOI: 10.3390/pharmaceutics14112520 - https://www.nature.com/articles/d41573-023-00098-6
